Methylsilane
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
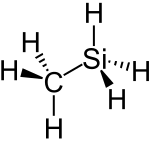
|
||||||||||||||||
| Wedges to clarify the spatial structure | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Methylsilane | |||||||||||||||
| other names |
Monomethylsilane |
|||||||||||||||
| Molecular formula | CH 6 Si | |||||||||||||||
| Brief description |
colorless gas with a foul odor |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 46.14 g mol −1 | |||||||||||||||
| Physical state |
gaseous |
|||||||||||||||
| density |
|
|||||||||||||||
| Melting point |
−156.8 ° C |
|||||||||||||||
| boiling point |
−57.5 ° C |
|||||||||||||||
| Vapor pressure |
|
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Methylsilane is a chemical compound from the group of organosilicon compounds .
presentation
Methylsilane can be produced in quantitative yield by reacting monochlorosilane with an excess of dimethylzinc .
properties
Methylsilane is an extremely flammable, colorless gas ( flash point ≤ −30 ° C) with a disgusting odor. It is heavier than air ( relative gas density = 1.606) and forms explosive mixtures with air (lower explosion limit at 1.3 % by volume or 25 g m −3 , upper explosion limit at 88.9% by volume or 1735 g · m −3 ) and can self-ignite in air. It decomposes when heated.
use
Methylsilane is used in chemical gas phase epitaxy (VPE) of germanium - doped silicon carbide layers (Ge: SiC).
Related links
Individual evidence
- ↑ a b c d e f g h i j entry to methylsilane in the GESTIS database of IFA , retrieved on July 2, 2018(JavaScript required) .
- ↑ Data sheet methylsilanes from Sigma-Aldrich , accessed on July 22, 2014 ( PDF ).
- ^ Alfred Stock, Carl Somieski: Siliciumwasserstoffe VI .: Chlorination and methylation of the monosilane . In: Reports of the German Chemical Society (A and B Series) . tape 52 , no. 4 , April 12, 1919, p. 695 , doi : 10.1002 / cber.19190520410 .
- ↑ Airliquide: Methylsilane


![{\ displaystyle \ mathrm {SiH_ {3} Cl + \ Zn (CH_ {3}) _ {2} \ {\ xrightarrow [{}] {}} \ Si (CH_ {3}) H_ {3} \ + \ ClZnCH_ {3}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/d610c5bc93f89cf12311b44109bbb3375010411b)