Tetramethylsilane
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
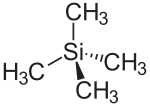
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Tetramethylsilane | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 4 H 12 Si | |||||||||||||||
| Brief description |
colorless liquid with a characteristic odor |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 88.23 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
0.65 g cm −3 |
|||||||||||||||
| Melting point |
−102 ° C (α-TMS) |
|||||||||||||||
| boiling point |
26 ° C |
|||||||||||||||
| Vapor pressure |
750 h Pa (20 ° C) |
|||||||||||||||
| solubility |
almost insoluble in water (20 mg l −1 at 25 ° C) |
|||||||||||||||
| Refractive index |
1.3587 (20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
Tetramethylsilane , with the constitutional formula Si (CH 3 ) 4 , (TMS) is a chemical substance that consists of a central silicon atom (Si) and four methyl groups (–CH 3 ) attached to it . TMS is largely chemically inert .
presentation
Tetramethylsilane can be synthesized from silicon tetrachloride and methyl magnesium chloride by the Grignard reaction . It can also be prepared by reacting chlorotrimethylsilane with sodium methylaluminum chloride.
One of the first syntheses of tetramethylsilane was achieved in 1865 by James Mason Crafts and Charles Friedel by reacting silicon tetrachloride with dimethylzinc at 200 ° C.
use
As a reference in NMR spectroscopy
TMS is used in NMR spectroscopy as a standard substance for calibrating the chemical shift of 1 H and 13 C spectra in organic solvents. A chemical shift of 0 ppm is assigned to the TMS signal. It is particularly well suited for this purpose for the following reasons:
- TMS delivers a single intense signal in each of the two spectra.
- Because of the low electronegativity of silicon, the signal is at a lower frequency than the signals of ordinary organic compounds .
- TMS is largely chemically inert and can therefore be added to the sample as an internal standard.
- TMS is highly volatile and can therefore be easily removed again after the measurement.
Alternatives
In aqueous solutions in which TMS is practically insoluble, the water-soluble derivatives DSS (2,2-dimethyl-2-silapentane-5-sulfonic acid, sodium salt) or TSP (3- (trimethylsilyl) -propionic acid, sodium salt) are used instead. In organic solvents, octamethylcyclotetrasiloxane (OMS) can be used as an alternative to TMS if a higher boiling point is desired.
As a methylating agent
Although - as stated above - TMS is largely inert, it has been used successfully as a methylating agent in transmetalation reactions :
safety instructions
The vapors are irritating to the eyes and the respiratory tract, in higher concentrations there is a narcotic effect.
Individual evidence
- ↑ a b c d e f g h Entry on tetramethylsilane in the GESTIS substance database of the IFA , accessed on February 17, 2017(JavaScript required) .
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Physical Constants of Organic Compounds, pp. 3-480.
- ↑ Georg Brauer (Ed.), With the collaboration of Marianne Baudler a . a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6 , p. 705.
- ↑ JM Crafts, C. Friedel: Ueber das Siliciummethyl und die Kieselsäure-Methylather . In: Annals of Chemistry and Pharmacy . tape 136 , no. 2 , 1865, p. 203-211 , doi : 10.1002 / jlac.18651360217 .
- ↑ H. Schmidbaur, W. Findeiss: A Simple Route to Organogallium Compounds . In: Angewandte Chemie International Edition in English . tape 3 , no. October 10 , 1964, p. 696 , doi : 10.1002 / anie.196406961 .




