Molybdenum (III) bromide
| Crystal structure | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
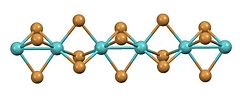
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Molybdenum (III) bromide | ||||||||||||||||||
| other names |
Molybdenum tribromide |
||||||||||||||||||
| Ratio formula | MoBr 3 | ||||||||||||||||||
| Brief description |
black solid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 335.70 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| density |
4.89 g cm −3 |
||||||||||||||||||
| Melting point |
500 ° C (decomposition) |
||||||||||||||||||
| solubility |
almost insoluble in water |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Molybdenum (III) bromide is an inorganic chemical compound of molybdenum from the group of bromides .
Extraction and presentation
Molybdenum (III) bromide can be obtained by reacting molybdenum with bromine in a carbon dioxide atmosphere at 350 ° C.
It is also possible to display it by reducing molybdenum (IV) bromide with molybdenum, hydrogen or a hydrocarbon.
properties
Molybdenum (III) bromide is a black solid that is in the form of densely matted crystal needles and is insoluble in water and acids, but easily soluble in boiling, anhydrous pyridine (as [MoBr 3 Py 3 ]). It decomposes during annealing in the absence of air into molybdenum (II) bromide and bromine. Molybdenum (III) bromide has an orthorhombic crystal structure of the ruthenium (III) bromide type with the space group Pnmm (space group no. 59, position 2) (a = 659.5 pm, b = 1142.3 pm, c = 606.0 pm) and four formula units in the unit cell .
Individual evidence
- ↑ a b c Georg Brauer (Ed.) U. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume III, Ferdinand Enke, Stuttgart 1981, ISBN 3-432-87823-0 , p. 1537.
- ↑ a b c W. M. Haynes, David R. Lide, Thomas J. Bruno: CRC Handbook of Chemistry and Physics 2012-2013 . CRC Press, 2012, ISBN 1-4398-8049-2 , pp. 4–76 ( limited preview in Google Book Search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ^ Dale L. Perry: Handbook of Inorganic Compounds, Second Edition . Taylor & Francis, 2011, ISBN 1-4398-1462-7 , pp. 279 ( limited preview in Google Book search).
- ↑ Dietrich Babel: The refinement of the MoBr3 structure. In: Journal of Solid State Chemistry . 4, 1972, pp. 410-416, doi : 10.1016 / 0022-4596 (72) 90156-9 .
