Sodium tartrate
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||
| L - (+) - disodium tartrate | |||||||||||||
| General | |||||||||||||
| Surname | Sodium tartrate | ||||||||||||
| other names |
|
||||||||||||
| Molecular formula | C 4 H 4 Na 2 O 6 | ||||||||||||
| Brief description |
colorless rhombic crystals (dihydrate) |
||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| Drug information | |||||||||||||
| ATC code | |||||||||||||
| properties | |||||||||||||
| Molar mass | 194.08 g · mol -1 | ||||||||||||
| Physical state |
firmly |
||||||||||||
| density |
1.81 g cm −3 (dihydrate) |
||||||||||||
| Melting point |
150 ° C (dihydrate) |
||||||||||||
| solubility |
good in water (330 g l −1 at 25 ° C for L - (+) - disodium tartrate) |
||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Sodium tartrate is a sodium salt of tartaric acid with the empirical formula C 4 H 4 O 6 Na 2 . It occurs in a bound form in many fruits.
Isomers
Like tartaric acid, sodium tartrate also has two stereocenters . Depending on the configuration of these centers, D - (-) - sodium tartrate [synonym: ( 2S , 3S ) -sodium tartrate ], L - (+) - sodium tartrate [synonym: ( 2R , 3R ) -sodium tartrate ] or the optically inactive meso - Sodium tartrate. In the meso form, one of the stereocenters is ( R ) - the other ( S ) -configured . "Sodium tartrate" without a specific name mostly refers to L - (+) - sodium tartrate.
| Isomers of sodium tartrate | |||
| Surname | L - (+) - sodium tartrate | D - (-) - sodium tartrate | meso sodium tartrate |
| Structural formula |
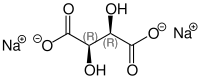
|
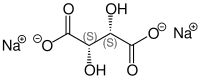
|

|
| other names | (2 R , 3 R ) -tartaric acid disodium salt | Disodium salt of (2 S , 3 S ) -tartaric acid | Disodium salt of meso -tartaric acid Disodium salt of (2 R , 3 S ) -tartaric acid (2 S , 3 R ) -tartaric acid disodium salt |
| CAS number | 868-18-8 (anhydrate) 6106-24-7 (dihydrate) |
109175-69-1 (anhydrate) 22476-07-9 (dihydrate) |
4504-50-1 |
| 51307-92-7 ( racemate ) | |||
| ? (Mixture of isomers) | |||
| EC number | 212-773-3 (anhydrate) 612-074-7 (dihydrate) |
244-213-9 (anhydrate) | 224-817-9 |
| ECHA info card |
100.011.613 (anhydrate) 100.122.993 (dihydrate) |
100.040.179 (anhydrate) | 100.022.563 |
| PubChem |
162637 (anhydrate) 12598458 (dihydrate) |
159905 (anhydrate) | 165240 |
| 13355 (mixture of isomers) | |||
| Wikidata |
Q27132338 (anhydrate) Q27276410 (dihydrate) |
Q27282569 (anhydrate) | Q27266651 |
| Q283499 (mixture of isomers) | |||
Presentation and extraction
Sodium tartrate is usually made from natural tartaric acid, which in turn is obtained from tartar from wine residues, which is first mixed with milk of lime and then with sulfuric acid .
use
Since a defined amount of water of crystallization is bound in crystalline sodium tartrate , it is a common primary standard for the Karl Fischer method , a widely used method for the quantitative determination of water by titration.
Sodium tartrate and sodium hydrogen tartrate from (natural) L - (+) - tartaric acid are used as leavening agents , as acidulants or acid regulators, and also as a substitute for table salt . Due to the property of tartrates to form stable complexes with iron and heavy metal ions, they support the effect of antioxidants and are therefore also used as complexing agents. It is approved in the EU as a food additive with the number E 335 with a permitted daily dose of 30 mg per kilogram of body weight and is then considered to be harmless.
Individual evidence
- ↑ Entry on E 335: Sodium tartrates in the European database for food additives, accessed on June 27, 2020.
- ↑ a b c Entry on sodium tartrate. In: Römpp Online . Georg Thieme Verlag, accessed on April 26, 2018.
- ↑ George WA Milne: Drugs: Definitions and Properties Definitions and Properties . Routledge, 2018, ISBN 978-1-351-78989-9 ( limited preview in Google Book Search).
- ↑ a b Sodium L-tartrate dibasic dihydrate data sheet from Sigma-Aldrich , accessed on April 26, 2018 ( PDF ).
