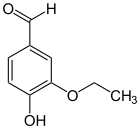
Novovanillin
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Novovanillin | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 9 H 10 O 3 | ||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 166.17 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
66-68 ° C |
||||||||||||||||||
| boiling point |
263-264 ° C |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Novovanillin ( 3-ethoxy-2-hydroxybenzaldehyde , ortho- ethylvanillin) is an organic chemical compound with the empirical formula C 9 H 10 O 3 . It is a derivative of benzaldehyde with an additional hydroxy and an ethoxy group . The hydroxy group is here, as in the ortho -Vanillin in ortho -position to the aldehyde group. In ethyl vanillin are these two groups in the para position.
Novovanillin is a solid, melting at 66–68 ° C and boiling at 263–264 ° C.
Tetravalent vanadium complexes (with VO 2+ ) of the formula [V (IV) O (dsal) 2 (H 2 O)] with ortho- hydroxy-substituted benzaldehydes are known (with Hdsal = salicylaldehyde , ortho-vanillin or novovanillin).
literature
- Elmar Profft : “To the knowledge of o-vanillin and novovanillin (= 2-oxy-3-ethoxybenzaldehyde-1). I “, in: Journal für Praktische Chemie , 1957 , 5 (3-4), pp. 175-181; doi : 10.1002 / prac.19570050303 .
- Elmar Profft, Peter Märker: “To the knowledge of o-vanillin and novovanillin (= 2-oxy-3-ethoxybenzaldehyde-1). IV “, in: Journal für Praktische Chemie , 1959 , 8 (3–4), pp. 199–206; doi : 10.1002 / prac.19590080311 .
- Elmar Profft: "Data on the ortho- & novovanillins (2-hydroxy-3-ethoxybenzaldehyde-1)", in: Arzneimittel-Forschung , 1959 , 9 (3), pp. 157-161; PMID 13651025 .
- Elmar Profft: “Data on o-vanillin & novovanillin (2-hydroxy-3-ethoxybenzaldehyde). II. Preparation of 2,3-dialkoxyphenyl-beta-ethylamine ", in: Archives of Pharmacy and Reports of the German Pharmaceutical Society , 1959 , 292 (2), pp. 70-75, PMID 13650594 .
- E. Profft, K. Stühmer: "Benzoins, benzils and benzilic acids of o-vanillin- and o-novovanillin ether", in: Archives of Pharmacy and Reports of the German Pharmaceutical Society , 1965 , 298 (10), p. 677– 685; PMID 5222289 .
Individual evidence
- ↑ a b c d e data sheet 3-ethoxysalicylaldehyde from Sigma-Aldrich , accessed on April 16, 2011 ( PDF ).
- ↑ JC Pessoa, I. Cavaco, I. Correia, I. Tomaz, T. Duarte, PM Matias: "Oxovanadium (IV) complexes with aromatic aldehydes", in: J Inorg Biochem , 2000 , 80 (1-2), p 35-39; PMID 10885461 .
Web links
- Entry on novovanillin . In: P. J. Linstrom, W. G. Mallard (Eds.): NIST Chemistry WebBook, NIST Standard Reference Database Number 69 . National Institute of Standards and Technology , Gaithersburg MD, accessed December 14, 2012.