Octabenzone
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
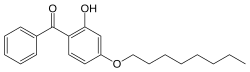
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Octabenzone | ||||||||||||||||||
| other names |
[2-hydroxy-4- (octyloxy) phenyl] (phenyl) methanone ( IUPAC ) |
||||||||||||||||||
| Molecular formula | C 21 H 26 O 3 | ||||||||||||||||||
| Brief description |
colorless to light yellow odorless solid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 326.43 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| density |
1.16 g cm −3 |
||||||||||||||||||
| Melting point |
47-49 ° C |
||||||||||||||||||
| boiling point |
175 ° C at 1.332 hPa |
||||||||||||||||||
| solubility |
|
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| Toxicological data | |||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Octabenzone is a chemical compound from the benzophenone group .
Extraction and presentation
Octabenzone can be obtained by reacting 2,4-dihydroxybenzophenone with n- octyl bromide or n- octyl chloride or by alkylating 2,4-dihydroxybenzophenone with an octyl halide.
properties
Octabenzone is a colorless to light yellow odorless solid, practically insoluble in water. It decomposes at temperatures above 300 ° C.
use
Octabenzone is an effective photostabilizer for a wide variety of plastics. It can be used in food packaging as an antioxidant and stabilizer and as a UV absorber in inks, coatings, adhesives, sealants, polyurethane and plastics.
Risk assessment
Octabenzon was included by the EU in 2013 in accordance with Regulation (EC) No. 1907/2006 (REACH) in the context of substance evaluation in the Community's rolling action plan ( CoRAP ). The effects of the substance on human health and the environment are re-evaluated and, if necessary, follow-up measures are initiated. The reasons for the inclusion of Octabenzone were concerns about consumer use , high (aggregated) tonnage and widespread use, as well as the suspected dangers of sensitizing properties and as a potential endocrine disruptor . The re-evaluation took place from 2013 and was carried out by Italy . A final report was then published.
Individual evidence
- ↑ a b c d e f g h Data sheet Octabenzon, 98% from Sigma-Aldrich , accessed on February 20, 2016 ( PDF ).
- ↑ a b c d e T.SS Dikshith: Hazardous Chemicals Safety Management and Global Regulations . CRC Press, 2013, ISBN 978-1-4398-7821-7 , pp. 297 ( limited preview in Google Book search).
- ↑ a b c Entry on Octabenzon in the Hazardous Substances Data Bank , accessed February 20, 2016.
- ↑ Entry on Octabenzon in the GESTIS substance database of the IFA , accessed on January 14, 2020(JavaScript required) .
- ↑ Robert Martin: Aromatic Hydroxyketones: Preparation and Physical Properties Vol.1: Hydroxybenzophenones Vol.2: Hydroxyacetophenones I Vol.3: Hydroxyacetophenones II Vol.4: Hydroxypropiophenones, Hydroxyisobutyrophenones, Hydroxypivalophenones and Derivatives . Springer Science & Business Media, 2011, ISBN 978-1-4020-9787-4 , pp. 135 ( limited preview in Google Book search).
- ↑ European Chemicals Agency (ECHA): Substance Evaluation Conclusion and Evaluation Report .
- ↑ Community rolling action plan ( CoRAP ) of the European Chemicals Agency (ECHA): Octabenzone , accessed on March 26, 2019.
