Oxaloacetic acid
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Oxaloacetic acid | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 4 H 4 O 5 | |||||||||||||||
| Brief description |
white to light yellow, crystalline solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 132.07 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
161 ° C ( decomposition ) |
|||||||||||||||
| solubility |
good in water (100 g l −1 ) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Oxaloacetic acid is an oxodicarboxylic acid and, as a metabolite of the citric acid cycle, an important node in the metabolism. Their salts are called oxalacetates (OA).
Occurrence
Oxaloacetic acid occurs in high concentrations in higher plants.
properties
As a dicarboxylic acid, oxaloacetic acid is both α- and β- keto acid and shows the reactions of both substance classes:
- as an α-keto acid, it is subject to nucleophilic attack
- it can be converted into the homologous amino acid aspartic acid by transamination at C-α ;
- In the initial reaction of the citric acid cycle , acetyl-CoA is the nucleophile due to its H-acidic methyl group , whereby citrate is formed;
- as β-keto acid, it shows the instability of this class of substances and goes through decarboxylation
- in a partial reaction catalyzed by the malate enzyme (ME) in pyruvate (Pyr) or
- in a reaction catalyzed by phosphoenolpyruvate carboxykinase (PEPCK) to phosphoenolpyruvate (PEP); the latter is an important partial reaction in gluconeogenesis ;
- Conversely, the use of ATP also enables the carboxylation of pyruvate to oxaloacetate by the biotin- dependent enzyme pyruvate carboxylase . This process is considered to be the most important anaplerotic (i.e. filling up the citric acid cycle ) reaction;
- Oxaloacetate can absorb 2 reduction equivalents per mole and transport it (after the transition to malate , mal) across the mitochondria (inner) membrane. Both directions of transport are possible:
- the reaction sequence OA → Mal || Mal → OA supplies reduction equivalents from the cytosol to the respiratory chain and serves to gain ATP ;
- the reverse running of the same reaction sequence transports reduction equivalents into the cytosol (e.g. with excess citrate). These can be used there in the form of NADPH, H + for the synthesis of fats . The substrate Acetyl-CoA (from citrate) is also available for this process .
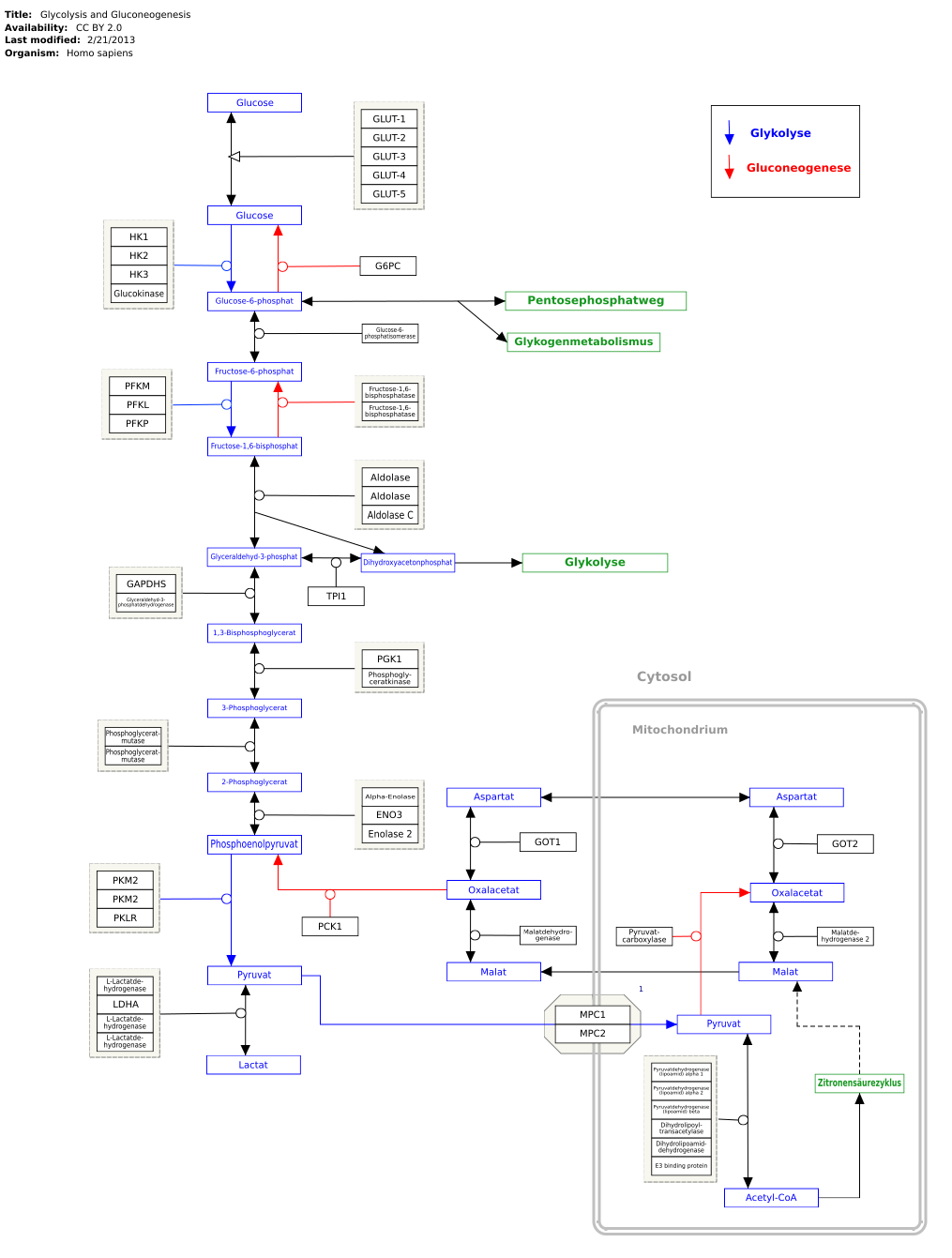
Glycolysis
Genes, proteins and metabolites are linked to the respective articles. The metabolic pathway can be edited at WikiPathways :
See also
Web links
Wiktionary: Oxaloacetic acid - explanations of meanings, word origins, synonyms, translations
Individual evidence
- ↑ Data sheet oxaloacetic acid from Acros, accessed on May 30, 2007.
- ↑ Data sheet oxaloacetic acid from AlfaAesar, accessed on February 20, 2010 ( PDF )(JavaScript required) .
- ↑ a b c data sheet Oxaloacetic acid from Sigma-Aldrich , accessed on March 3, 2020 ( PDF ).