Phosphorylase kinase
| Phosphorylase kinase | ||
|---|---|---|
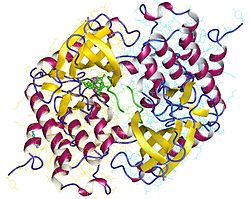
|
||
| Phosphorylase kinase (fragment) dimer, Oryctolagus cuniculus according to PDB 2PHK | ||
| Mass / length primary structure | 10996 = 4 * (1223 + 1092 + 386 + 148) amino acids | |
| Secondary to quaternary structure | (αβγδ) 4 | |
| Isoforms | α 2 (1235 aas); γ 2 (406 carrion) |
|
| Identifier | ||
| Gene name (s) | PHKA1 , PHKA2 , PHKB , PHKG1 , PHKG2 | |
| Enzyme classification | ||
| EC, category | 2.7.11.19 , Transferase | |
| Response type | Phosphorylation | |
| Substrate | 2 ATP + phosphorylase b | |
| Products | 2 ADP + phosphorylase a | |
| Occurrence | ||
| Parent taxon | Animals, bacteria | |
Phosphorylase kinase is an enzyme that phosphorylates the enzyme glycogen phosphorylase and thus activates it. As a result, it plays a central role in the cascading activation mechanism of glycogen breakdown in animals and bacteria. It is the first serine / threonine kinase that was discovered in 1959 by Edwin Gerhard Krebs and his colleagues.
construction
Phophorylase kinase is a very large protein with a mass of 1200 kD . It has four subunits ; in skeletal muscle there is a subunit structure of (αβγδ) 4 . The β-subunit has a catalytic activity, the other subunits fulfill regulatory functions. The δ subunit is calmodulin .
regulation
The enzyme is regulated by two processes: by reversible phosphorylation and by allosteric regulation.
In the former, protein kinase A (PKA) plays a crucial role. This transfers a phosphate group to the β-subunit of the initially weakly active phosphorylase kinase. As a result, the activity level of the kinase is increased. The PKA itself is activated from an inactive form by a second messenger , cAMP . One reason for cAMP release is the presence of hormones such as adrenaline and glucagon .
The phosphorylase kinase can, however, also be activated in a partially active form by an increased Ca 2+ level. Calmodulin , which corresponds to the δ-subunit of the enzyme, serves as the calcium sensor .
If both the β-subunit of the phosphorylase kinase is in a phosphorylated form and the calmodulin has bound calcium ions in the δ-subunit, the enzyme is fully active.
function
The glycogen breakdown is triggered by a hunger or stress signal via a cascade signal transmission path. After binding the hormone adrenaline (stress signal ) or glucagon (hunger signal), cAMP is released as a second messenger , which in turn activates protein kinase A. As described above, this phosphorylates phosphorylase kinase. When activated, the latter in turn phosphorylates the inactive glycogen phosphorylase, a dimeric enzyme. Inactivated it is also known as phosphorylase b , after phosphorylation it is converted into an active form (phosphorylase a ). The activated glycogen phosphorylase can enzymatically break down glycogen to glucose-1-phosphate and then release it into the blood in the form of glucose in further reaction steps .
Phosphorylase kinase thus plays a central role in the signal transduction pathway for glycogen breakdown.
See also
literature
- Berg, Tymoczko, Stryer: Biochemistry . 5th edition. Spektrum Akademischer Verlag, Heidelberg 2003, ISBN 3827413036

