Acid anhydrides
Acid anhydrides (mostly just called anhydrides ) is the name for a group of chemical compounds that are always derivatives of acids containing oxygen . Formally, they arise from the splitting off of water (dehydration) from an acid. Inorganic oxygen acids form oxides such as B. from phosphoric acid phosphorus pentoxide . So-called carboxylic acid anhydrides are formed from the organic carboxylic acids .
Types
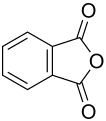
In the case of organic acids ( carboxylic acids ), two acid groups are linked via an oxygen bridge in the molecule . The two acid groups can either come from one molecule ( intramolecular ) as in phthalic anhydride or belong to two different molecules ( intermolecular ) as in acetic anhydride . In the case of organic acids, the acid anhydride is named by adding the ending -anhydride to the name of the acid.
In the case of inorganic acids, the substances usually have their own names. Phosphorus pentoxide is e.g. B. the acid anhydride of phosphoric acid . Mixed anhydrides are formed by splitting off water from two different acids.
|
|
||||||||||||||||||||||
presentation
Acid anhydrides can be produced in a number of ways:
- By reaction of a carboxylic acid chloride with the salt of a carboxylic acid or
- by splitting off water using a strong dehydrating agent e.g. B. sulfuric acid .
- Special anhydrides can also be produced in other ways. So z. B. maleic anhydride obtained by oxidation of n -butane or (obsolete) benzene and phthalic anhydride from o -xylene or naphthalene .
- Carbon monoxide is a special case ; it can be produced by dehydrating formic acid and introducing carbon monoxide into caustic soda creates formate . Thus, an inorganic substance is the acid anhydride of an organic acid.
Properties and use
Acid anhydrides react exothermically with water to form the corresponding acid (s). Acid anhydrides can be used for halogen-free Friedel-Crafts acylation or for drying the corresponding acid.
Other mixed anhydrides
Mixed anhydrides result from the condensation of two different acids. Formally, carboxylic acid halides are anhydrides of a carboxylic acid with a hydrohalic acid z. B. Acetyl Chloride :
Carboxylic acid halides or other anhydrides formed formally by the reaction of an acid with a carboxylic acid, however, often form their own substance classes and are not referred to as anhydrides.