Tetrafluoromethane
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
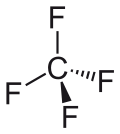
|
||||||||||||||||
| Wedges to clarify the geometry | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Tetrafluoromethane | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | CF 4 | |||||||||||||||
| Brief description |
odorless and colorless gas |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 88.01 g mol −1 | |||||||||||||||
| Physical state |
gaseous |
|||||||||||||||
| density |
3.72 kg m −3 (15 ° C, 1013 mbar) |
|||||||||||||||
| Melting point |
−184 ° C |
|||||||||||||||
| boiling point |
−128 ° C |
|||||||||||||||
| solubility |
heavy in water (20 mg l −1 at 20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Global warming potential |
7349 (based on 100 years) |
|||||||||||||||
| Thermodynamic properties | ||||||||||||||||
| ΔH f 0 |
−933.6 kJ / mol |
|||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Tetrafluoromethane (also carbon tetrafluoride ) is a chemical compound from the series of fluorocarbons . With him, all hydrogen atoms of methane are substituted by fluorine atoms . Both names are according to the IUPAC - Nomenclature correctly, depending on whether the connection as organic ( tetrafluoromethane ) or inorganic ( carbon tetrafluoride ) compound is considered.
presentation
Tetrafluoromethane can be produced on the one hand by burning carbon in fluorine , on the other hand by electrical discharge in a carbon monoxide-fluorine mixture:
Another method is the conversion of silicon carbide with fluorine to tetrafluoromethane and silicon tetrafluoride :
The conversion is almost quantitative and the gaseous silicon compound can be removed from the gas mixture by washing with a sodium hydroxide solution ( hydrolysis ).
properties

Physical Properties
Tetrafluoromethane is a colorless and odorless gas with a melting point of −184 ° C and a boiling point of −128 ° C. Tetrafluoromethane is very poorly soluble in water (20 mg per kg of water at 20 ° C), somewhat in ethanol (approx. 80 mg per kg of ethanol at 25 ° C) and benzene (approx. 64 mg per kg of benzene at 25 ° C) better.
Chemical properties
Tetrafluoromethane is very inert and is not attacked by acids and alkalis. Only boiling alkali metals and hot alkali metal vapors can slowly attack it. Thermal decomposition above 1000 ° C produces toxic substances ( carbonyl fluoride , carbon monoxide ), and in the presence of water also the aggressive hydrogen fluoride.
It has a global warming potential of 7349, making it an extremely powerful greenhouse gas .
Use and emissions
Tetrafluoromethane is used as a refrigerant to replace CFCs , as it does not deplete the ozone layer. Due to the high global warming potential, however, the F-gas regulation stipulates that systems with the corresponding gases should leak as little amounts as possible. Large amounts of tetrafluoromethane are released during aluminum production.
toxicology
Tetrafluoromethane is considered non-toxic. However, it can cause asphyxiation in closed spaces by displacing the air. A narcotic effect is possible at lower concentrations.
Individual evidence
- ↑ a b c d e Entry on tetrafluoromethane in the GESTIS substance database of the IFA , accessed on February 1, 2016(JavaScript required) .
- ↑ a b c d Datasheet Tetrafluoromethane at Air Liquide.
- ↑ a b B. A. Cosgrove, J. Walkley: Solubilities of gases in H 2 O and 2 H 2 O. in: J. Chromatogr. A 216, 1981, pp. 161-167; doi: 10.1016 / S0021-9673 (00) 82344-4 .
- ↑ a b G. Myhre, D. Shindell et al .: Climate Change 2013: The Physical Science Basis . Working Group I contribution to the IPCC Fifth Assessment Report. Ed .: Intergovernmental Panel on Climate Change . 2013, Chapter 8: Anthropogenic and Natural Radiative Forcing, pp. 24-39; Table 8.SM.16 ( PDF ).
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Standard Thermodynamic Properties of Chemical Substances, pp. 5-19.
- ↑ G. Brauer (Ed.): Handbook of Preparative Inorganic Chemistry. 2nd Edition, Volume 1, Academic Press 1963, pp. 203-204.
- ↑ a b Homer F. Priest: Anhydrous metal fluorides . In: Ludwig F. Audrieth (Ed.): Inorganic Syntheses . tape 3 . McGraw-Hill, Inc., 1950, pp. 171-183 (English).
- ↑ Shiqing Bo, Rubin Battino, Emmerich Wilhelm: The Solubility of Gases in Liquids. 19. The Solubility of He, Ne, Ar, Kr, Xe, CH 4 , CF 4 , SF 6 in normal 1-alkanols nC I H 2I + 1 OH (1 ≤ I ≤ 11) at 298.15 K. In: J. Chem. Eng. Data Vol. 38, No. 4, 1993, pp. 611-616; doi: 10.1021 / je00012a035 .
- ↑ Graham Archer, Joel H. Hildebrand: The Solubility and Entropy of Solution of carbon tetrafluoride and sulfur hexafluoride in nonpolar Solvents. In: J. Phys. Chem. Vol. 67, No. 9, 1963, pp. 1830-1833; doi: 10.1021 / j100803a021 .



