Thulium (III) oxide
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
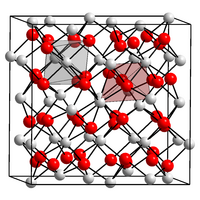
|
||||||||||||||||
| __ Tm 3+ __ O 2− | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Thulium (III) oxide | |||||||||||||||
| other names |
|
|||||||||||||||
| Ratio formula | Tm 2 O 3 | |||||||||||||||
| Brief description |
white to slightly greenish solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 385.87 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
8.6 g cm −3 |
|||||||||||||||
| Melting point |
2341 ° C |
|||||||||||||||
| boiling point |
3945 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Thulium (III) oxide is a chemical compound from the group of oxides .
history
Thulium (III) oxide was discovered in 1879 by the Swedish chemist Per Teodor Cleve together with holmium in erbia ( erbium oxide ) (see history of holmium ) after he had already determined the presence of several impurities in it in 1874.
Occurrence
Thulium (III) oxide occurs in very low concentrations in the minerals monazite (0.002%) and bastnasite (0.0008%). The world production of thulium in the form of thulium oxide is about 50 tons per year.
Extraction and presentation
Thulium (III) oxide can be obtained by burning thulium in air.
It can also be obtained by thermal decomposition of thulium oxalate , thulium carbonate or thulium acetate at temperatures around 700 ° C.
properties
Thulium (III) oxide is a white to slightly greenish solid. It has a cubic crystal structure.
use
Thulium (III) oxide changes the optical properties of glass. Tm- doped glass fiber is used for 1.8 µm lasers .
Individual evidence
- ↑ a b American Elements: Thulium Oxide
- ↑ a b c David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Properties of the Elements and Inorganic Compounds, pp. 4-95.
- ↑ a b c data sheet Thulium (III) oxide, 99.99% trace metals basis from Sigma-Aldrich , accessed on March 7, 2012 ( PDF ).
- ^ A b John Emsley: Nature's building blocks: an AZ guide to the elements . Oxford University Press, 2001, ISBN 0-19-850341-5 , pp. 442–443 ( limited preview in Google Book Search).
- ↑ Web elements: Thulium (III) oxide
- ^ Carl Alfred Jacobson, Clifford A. Hampel: Encyclopedia of chemical reactions, Volume 7 . Reinhold Pub. Corp., 1946, OCLC 660994317 , p. 318 ( limited preview in Google Book search).
- ↑ Gerd Meyer, Lester R. Morss: Synthesis of lanthanide and actinide compounds . Springer Netherlands, 1990, ISBN 0-7923-1018-7 , pp. 196 ( limited preview in Google Book search).
- ↑ Ginya Adachi, Nobuhito Imanaka, Zhen Chuan Kang: Binary rare earth oxides . Springer Netherlands, 2004, ISBN 1-4020-2568-8 , pp. 95 ff., 151 ( limited preview in Google Book search).
- ↑ A. de Pablos-Martín, D. Ristic, S. Bhattacharyya, Th. Höche, GC Mather, M. O Ramírez, S. Soria, M. Ferrari, GC Righini, LE Bausá, A. Durán, MJ Pascual, J McKittrick: Effects of Tm 3+ Additions on the Crystallization of LaF 3 Nanocrystals in Oxyfluoride Glasses: Optical Characterization and Up-Conversion . In: Journal of the American Ceramic Society . tape 96 , no. 2 , 2013, p. 447-457 , doi : 10.1111 / jace.12120 (English).
- ↑ 1-kW, All-Glass Tm: fiber laser ( Memento of the original from March 10, 2016 in the Internet Archive ) Info: The archive link was inserted automatically and has not yet been checked. Please check the original and archive link according to the instructions and then remove this notice. , 2010


