Tosylaminohexanoic acid
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Tosylaminohexanoic acid | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 13 H 19 NO 4 S | ||||||||||||||||||
| Brief description |
white crystalline powder |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 285.36 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point | |||||||||||||||||||
| solubility |
readily soluble in water as a triethanolamine salt |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Tosylaminohexanoic acid combines structural elements of a sulfonamide and an amino acid and is obtained by reacting ε-aminocaproic acid with para-toluenesulfonic acid chloride . It is used, especially in the form of its triethanolamine salt, as a corrosion protection agent in aqueous systems.
presentation
The reaction of 6-aminohexanoic acid with tosyl chloride in an alkaline (pH approx. 11) water / 1,4-dioxane mixture produces tosylaminohexanoic acid after acidification, which can be recrystallized from 40% ethanol for purification .
or a solution of tosyl chloride in tetrahydrofuran is added to a solution of ε-aminocaproic acid in 1M sodium hydroxide solution and reacted at pH 11 to form p -toluenesulfonyl-ε-aminocaproic acid in 94% yield.
properties
Tosylaminohexanoic acid is a white crystalline solid which is hardly soluble in water at a neutral pH value and readily soluble in alkaline pH. The acid has a low water hazard potential. and is very compatible with other corrosion inhibitors .
use
6- (N-Tosylamino) caproic acid is a so-called ash-free and low-foaming inhibitor for the corrosion of iron and iron alloys in aqueous systems and is mostly used as a triethanolamine salt in concentrations of 0.2 to 2.5% active ingredient content.
Tosylaminohexanoic acid is also a highly effective corrosion inhibitor in systems with several metals. The compound - especially as a salt with triethanolamine, which also has anti-corrosive properties - reduces the corrosiveness of water-containing cooling and hydraulic fluids , metalworking and cleaning fluids and cooling lubricants such as drilling and cutting emulsions (English metal working fluids ) and provides corrosion protection during wire drawing as well as for offshore applications.
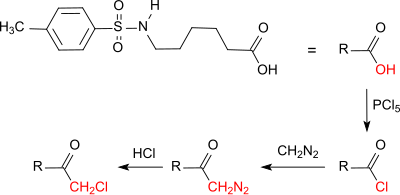
The chloromethyl ketone derived from tosylaminohexanoic acid
showed high antineoplastic activity in the animal model Ehrlich Ascites Carcinoma (experimental mouse tumor).
Individual evidence
- ↑ a b c B. Benacerraf, BB Levine: Immunological specificity of delayed and immediate hypersensitivity reactions . In: J. Exptl. Med. Band 115 , no. 5 , 1962, pp. 1023-1036 , doi : 10.1084 / jem.115.5.1023 , PMC 2137528 (free full text).
- ↑ a b V.H. Pavlidis, ED Chan, L. Pennington, M. McParland, M. Whitehead, IGC Coutts: Synthesis of aliphatic amines related to monodansylcadaverine via the oxidative rearrangement of aliphatic amides with phenyl iodosyl bis (trifluoroacetate) . In: Synth. Commun. tape 18 , no. 14 , 1988, pp. 1615-1624 , doi : 10.1080 / 00397918808081322 .
- ↑ Patent US3788991 : Metal working agents. Registered on January 24, 1972 , published on January 29, 1974 , applicant: Farbwerke Hoechst AG, inventor: H. Diery, S. Rittner, H. Lorke.
- ↑ a b Matrix Scientific: 6 - {((4-Methylphenyl) sulfonyl) amino} hexanoic acid , accessed December 27, 2019.
- ↑ a b c Metall-Chemie, product data sheet TC® 50, ( online ).
- ↑ Patent US8759264B2 : Water-based hydraulic fluids comprising dithio-di (aryl carboxylic acids). Registered on December 2, 2009 , published on June 24, 2014 , applicant: Clariant Finance (BVI) Ltd., inventor: R. Kupfer, C. Cohrs.
- ↑ Patent US20120088706A1 : Water-mixed metal working fluids containing ether pyrrolidone carboxylic acids. Registered on May 19, 2010 , published on April 12, 2012 , applicant: Clariant Finance (BVI) Ltd., inventors: R. Kupfer, C. Cohrs, A. Roesch.
- ↑ Patent WO0031218 : Metalworking and cleaning processes. Registered on November 17, 1999 , published on June 1, 2000 , applicant: Henkel KGaA, inventor: J. Geke, B. Stedry, W. Klose.
- ↑ Patent US20120088705A1 : Polyalkylene glycol-based ether pyrrolidone carboxylic acids, and concentrates for the production of synthetic cooling lubricants containing the same. Registered on May 19, 2010 , published on April 12, 2012 , applicant: Clariant Finance (BVI) Ltd., inventors: R. Kupfer, C. Cohrs, A. Roesch.
- ↑ Z. Sajadi, M. Kashani, LJ Loeffler, IH Hall: Antitumor agents: Diazomethyl ketone and chloromethyl ketone analogues prepared from N-tosyl amino acids . In: J. Med. Chem. Volume 23 , no. 3 , 1980, p. 275-278 , doi : 10.1021 / jm00177a012 .
- ↑ M. Ozaslan, ID Karagoz, IH Kilic, ME Guldur: Review: Ehrlich ascites carcinoma . In: Afr. J. Biotechnol. tape 10 , no. 13 , 2011, p. 2375-2378 , doi : 10.5897 / AJBx10.017 .