2-methoxyethyl mercury acetate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
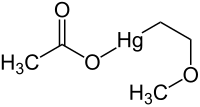
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | 2-methoxyethyl mercury acetate | |||||||||||||||
| other names |
Panogen Metox |
|||||||||||||||
| Molecular formula | C 5 H 10 HgO 3 | |||||||||||||||
| Brief description |
white crystals |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 318.72 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
40-42 ° C |
|||||||||||||||
| solubility |
soluble in water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
2- Methoxyethylmercury acetate , also known under its trade name Panogen Metox , is an extremely toxic organometallic compound of mercury . It was used for dressing seeds and replaced the older Panogen .
Extraction and presentation
2-Methoxyethyl mercury acetate can be obtained by oxymercuration of ethene with mercury (II) acetate and methanol .
Admission
2-Methoxyethylmercury acetate is not approved as a crop protection agent in the European Union or Switzerland .
Individual evidence
- ↑ a b c d e Entry on 2-methoxyethylmercuric acetate in the GESTIS substance database of the IFA , accessed on September 2, 2013(JavaScript required) .
- ↑ Thomas A. Unger: Pesticide Synthesis Handbook . William Andrew, 1996, ISBN 0-8155-1853-6 , pp. 395 ( limited preview in Google Book search).
- ^ Directorate-General for Health and Food Safety of the European Commission: EU pesticide database ; Entry in the national directory of plant protection products in Switzerland ; Retrieved June 26, 2016.


