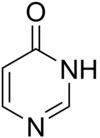
4-pyrimidinone
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | 4-pyrimidinone | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 4 H 4 N 2 O | ||||||||||||||||||
| Brief description |
white solid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 96.09 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
164-167 ° C |
||||||||||||||||||
| solubility |
soluble in water |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
4-pyrimidinone is a chemical compound from the group of pyrimidines . The compound is part of the basic structure of the nucleobase isocytosine . It is also found as a partial structure in mizolastine .
Extraction and presentation
4-Pyrimidinones can be obtained, among other things, by reacting a β-ketoester with thiourea , followed by desulfurization of the resulting product using Raney nickel .
properties
4-pyrimidinone is a white solid. In the spectrum of the compound instructions exist on an amide - tautomer .
Individual evidence
- ↑ a b c d e f g Data sheet 4-Pyrimidinone, 98% from AlfaAesar, accessed on August 9, 2014 ( PDF )(JavaScript required) .
- ↑ Patent application EP0326389A2 : Process for preparing 4-hydroxypyrimidine. Applied January 26, 1989 , published August 2, 1989 , Applicant: Ube Industries , Inventor: Kikuo C / O Ube Research Laboratory Ataka, Kiyoshi C / O Ube Research Laboratory Omori.
- ↑ Eberhard Breitmaier, Günther Jung: Organic chemistry . Georg Thieme Verlag, 2005, p. 641 ( limited preview in Google Book search).