Bis (4-chlorophenyl) sulfone
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
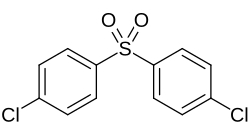
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Bis (4-chlorophenyl) sulfone | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 12 H 8 Cl 2 O 2 S | ||||||||||||||||||
| Brief description |
white solid with a faint odor |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 287.16 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| density |
1.504 g cm −3 |
||||||||||||||||||
| Melting point |
144-148 ° C |
||||||||||||||||||
| boiling point |
250 ° C (13 hPa) |
||||||||||||||||||
| solubility |
practically insoluble in water (0.86 mg l −1 at 20 ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Bis (4-chlorophenyl) sulfone is a chemical compound from the group of organic sulfones .
Extraction and presentation
Bis (4-chlorophenyl) sulfone can be produced by reacting chlorobenzene with a mixture of sulfur trioxide and dimethyl or diethyl pyrosulfate. The synthesis from chlorobenzene and chlorobenzenesulfonic acid is also described. This requires temperatures of 220 to 260 ° C, so that work must be carried out at high pressure.
The best-known method for the production of 4,4'-dichlorodiphenyl sulfone is the reaction of 4-chlorobenzenesulfone sulfonyl chloride with chlorobenzene, which takes place after a Friedel-Krafts reaction , whereby iron (III) chloride is used as a catalyst .
Processes for the preparation from chlorobenzene, sulfur trioxide or chlorosulfonic acid and thionyl chloride or phosgene without a catalyst are also known.
properties
Bis (4-chlorophenyl) sulfone is a flammable, hardly inflammable, white solid with a faint odor, which is practically insoluble in water. The compound has a monoclinic crystal structure with the space group I 2 / a (space group no. 15, position 3) .
use
Bis (4-chlorophenyl) sulfone is used as a precursor in the production of 4,4'-diaminodiphenyl sulfone and polysulfone plastics. It is also for the production of temperature-resistant polymers , such as PES or polysulfone (Udel) by polymerization with bisphenol A and bisphenol S used.
literature
- RS Chhabra, RA Herbert, JR Bucher, GS Travlos, JD Johnson, MR Hejtmancik: Toxicology and Carcinogenesis Studies of p, p'-Dichlorodiphenyl Sulfone in Rats and Mice. In: Toxicological Sciences. 60, 2001, p. 28, doi : 10.1093 / toxsci / 60.1.28 .
Individual evidence
- ↑ a b c d e f g h i Entry on bis (4-chlorophenyl) sulfone in the GESTIS substance database of the IFA , accessed on August 9, 2018(JavaScript required) .
- ↑ a b c Google Patents: DE3704932A1 - Process for the production of 4,4'-dichlorodiphenyl sulfone - Google Patents , accessed on August 9, 2018.
- ↑ JG Sime, SC Abrahams: The crystal and molecular structure of 4,4'-dichlorodiphenyl sulphone. In: Acta Crystallographica. 13, 1960, p. 1, doi : 10.1107 / S0365110X60000017 .
- ↑ Data sheet bis (4-chlorophenyl) sulfone, 99% from AlfaAesar, accessed on August 9, 2018 ( PDF )(JavaScript required) .
