Cerium (III) chloride
| Crystal structure | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||
| __ Ce 3+ __ Cl - | ||||||||||
| Crystal system | ||||||||||
| Space group |
P 6 3 / m (No. 176) |
|||||||||
| Coordination numbers |
Ce [9], Cl [3] |
|||||||||
| General | ||||||||||
| Surname | Cerium (III) chloride | |||||||||
| other names |
|
|||||||||
| Ratio formula | CeCl 3 | |||||||||
| Brief description |
white, hygroscopic, odorless solid |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 246.48 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| density |
3.92 g cm −3 (20 ° C) |
|||||||||
| Melting point |
848 ° C |
|||||||||
| boiling point |
1727 ° C |
|||||||||
| solubility |
easily in water (486 g l −1 at 25 ° C) |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| Toxicological data | ||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Cerium (III) chloride is a chemical compound of cerium and chlorine .
properties
It quickly absorbs water in moist air and forms various hydrates such as cerium (III) chloride heptahydrate (CeCl 3 · 7 H 2 O). This crystallizes triclinic , space group P 1 (space group no.2 ) , with the lattice parameters a = 790.2 pm , b = 821.0 pm, c = 918.8 pm, α = 70.52 °, β = 73, 14 ° and γ = 81.66 °.
use
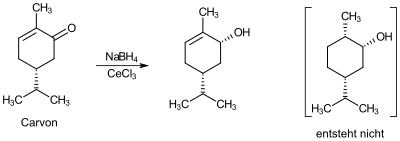
Cerium (III) chloride can be used as a starting material for the synthesis of other cerium compounds. It can also serve as a Lewis acid , for example as a catalyst for Friedel-Crafts acylations and for the alkylation of ketones . It can also be used for Luche reduction and as a component of detector crystals.
Web links
- Nobuhiro Takeda, Tsuneo Imamoto: Use of Cerium (III) chloride in the Reactions of Carbonyl Compounds with Organolithiums or Grignard Reagents for the Suppression of Abnormal Reactions: 1-Butyl-1,2,3,4-tetrahydro-1-naphthol In: Organic Syntheses . 76, 1999, p. 228, doi : 10.15227 / orgsyn.076.0228 ( PDF ).
Individual evidence
- ↑ a b c d e f g h Entry on cerium (III) chloride in the GESTIS substance database of the IFA , accessed on January 8, 2018(JavaScript required) .
- ↑ W. Levason, M. Webster: Di-μ-chloro-bis [heptaaquacerium (III)] tetrachloride. In: Acta Crystallographica , E58, 2002, pp. I76-i78, doi: 10.1107 / S1600536802014794 .
- ↑ Hintermann, Lukas: Catalytic enantioselective fluorination , dissertation, ETH Zurich, 2000. doi: 10.3929 / ethz-a-004062829
- ↑ Saint-Gobain: BrilLanCe ™ 380 crystal ( Memento of the original from June 9, 2012 in the Internet Archive ) Info: The archive link was inserted automatically and has not yet been checked. Please check the original and archive link according to the instructions and then remove this notice.