Energy converter
An energy converter exchanges energy between a system and the environment in at least two forms of energy . For example, a gasoline engine converts chemical energy into kinetic energy . Large-scale energy conversion systems such as power plants consist of several energy converters that gradually convert primary forms of energy into technically usable forms of energy such as electrical energy or thermal energy (process and district heating).
Energy conversion is called a category of processes in which energy is exchanged between a system and the environment in at least two forms of energy. The colloquial term energy generation is also common for energy conversion into electrical energy and refers to the form of energy made available after the process ( electrical energy ), see electricity generation .
history
Basics
Energy conversions are subject to physical laws. The energy in closed systems is a conservation quantity, so it can neither be generated nor destroyed. When it comes to technical use, the conversion efficiency is crucial, since in real systems it is not possible to convert 100% of one form of energy into another. There are always losses in other channels, mostly in the form of unused heat, i.e. thermal energy.
Both forms of energy do not carry any entropy in an ideal way of thinking, so that conversion losses, mostly heat, prevent a perpetual motion machine with absolute certainty. The entropy associated with this heat and generated in the process ensures the increase in total entropy required by the Second Law of Thermodynamics in real processes.
Examples
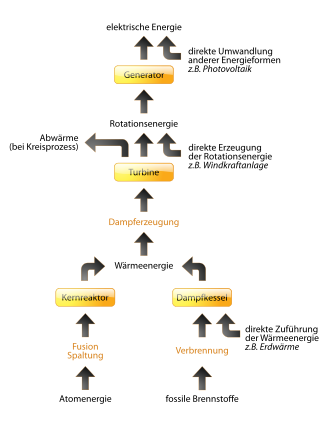
Almost all technical and biological processes are connected with the conversion of energy. Hence there are examples of energy converters for almost all pairs of forms of energy.
Electric motor
An electric motor converts electrical energy into kinetic energy .
Steam turbine
A steam turbine drives an electrical generator; thermal energy is converted into electrical energy. The turbine at the temperature T 1 of heat supplied .DELTA.Q 1 transmits the entropy .DELTA.S 1 = .DELTA.Q 1 / T 1 with it. The generated electrical energy ΔW has no entropy. If all the heat were converted into electrical energy, the entropy ΔS 1 would disappear, which would contradict the Second Law. The turbine must therefore emit waste heat ΔQ 2 with temperature T 2 , which has at least entropy ΔS 1 . The following applies for the energy: ΔQ 1 = ΔW + ΔQ 2 and for the entropy: ΔS 2 ≥ ΔS 1 ⇔ ΔQ 2 / T 2 ≥ ΔQ 1 / T 1 . From the second equation it follows that ΔQ 2 Δ ΔQ 1 · T 2 / T 1 . These waste heat losses .DELTA.Q 2 are absolutely necessary because of the second law and can not be undershot by any technical measures at given temperatures T 1 and T 2 . This limit of efficiency for a heat engine is realized in theoretical cycle processes such as the Carnot process . In addition, there are technical conversion losses.
solar power
The efficiency of conversions increases with the temperature differences (or their equivalent) that can be used in the conversion system. For example, the photoelectric effect is increasingly used in photovoltaics . The efficiencies achieved by direct photoelectric conversion are still below the conventional, double thermo-mechanical-electrical conversion. Much higher temperature differences occur in solar thermal power plants , in which, for example, the radiation energy concentrated by mirrors is first converted into thermal energy by absorption , then conventionally into mechanical and finally electrical energy.
|
after →
↓ from
|
Mechanical energy | Thermal energy | Radiant energy | Electrical power | Chemical energy | Nuclear energy |
|---|---|---|---|---|---|---|
| Mechanical energy | transmission | Brakes | Synchrotron radiation | generator | Reactions in the particle accelerator | |
| Thermal energy | Steam turbine | Heat exchanger | Black body | Thermocouple | Blast furnace | Supernova |
| Radiant energy | Radiometer | Solar collector | Nonlinear optics | Solar cell | photosynthesis | Core photo effect |
| Electrical power | Electric motor | Electric stove | lightning | transformer | accumulator | |
| Chemical energy | muscle | Oil heating | Firefly | Fuel cell | Coal gasification | |
| Nuclear energy | Atomic bomb | Nuclear reactor | gamma rays | Radionuclide battery | Radiolysis | Breeder |