Erythrose-4-phosphate
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
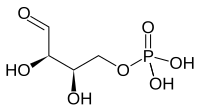
|
|||||||||||||
| General | |||||||||||||
| Surname | Erythrose-4-phosphate | ||||||||||||
| other names |
E4P |
||||||||||||
| Molecular formula | C 4 H 9 O 7 P | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 200.08 g mol −1 | ||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Erythrose-4-phosphate is a phosphoric acid ester of erythrose . It is an intermediate in the pentose phosphate route (PPP).
Reactions
In carbohydrate metabolism erythrose 4-phosphate is an important intermediate. It is formed in the course of the pentose phosphate pathway under the action of the enzyme transaldolase from sedoheptulose-7-phosphate and glyceraldehyde-3-phosphate , together with fructose-6-phosphate . Plants and many microorganisms use erythrose-4-phosphate together with phosphoenolpyruvate (PEP) in the shikimic acid pathway for the synthesis of the aromatic amino acids tryptophan , tyrosine and phenylalanine . In addition, erythrose-4-phosphate can be used in plants, bacteria and fungi for the synthesis of pyridoxine , the vitamin B6.
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ^ Hermann PT Ammon (ed.): Hunnius pharmaceutical dictionary. 9th, revised and expanded edition. Walter de Gruyter, Berlin et al. 2004, ISBN 3-11-017475-8 .
- ↑ Jeremy M. Berg, John L. Tymoczko, Lubert Stryer: Biochemistry. 6th edition. Spektrum Akademischer Verlag, Elsevier GmbH, Munich 2007, ISBN 978-3-8274-1800-5 , pp. 644-645, 773-775.