Europium (II) bromide
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| __ Eu 2+ __ Br - | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Europium (II) bromide | |||||||||||||||
| other names |
Europium dibromide |
|||||||||||||||
| Ratio formula | EuBr 2 | |||||||||||||||
| Brief description |
white solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 311.77 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
683 ° C |
|||||||||||||||
| boiling point |
1230 ° C |
|||||||||||||||
| solubility |
soluble in water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Europium (II) bromide is an inorganic chemical compound of europium from the group of bromides .
Extraction and presentation
Europium (II) bromide can be obtained by reducing europium (III) bromide with hydrogen at 350 ° C.
The representation by thermal decomposition of europium (III) bromide at 200 ° C is also possible.
The reaction of europium with ammonium bromide in liquid ammonia at −78 ° C is also possible. First of all, the ammonia from europium (II) bromide is obtained, which can then be broken down at around 200 ° C in a high vacuum.
properties
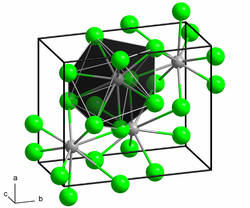
Europium (II) bromide is a white solid. The connection is extremely hygroscopic and can only be stored and handled under carefully dried protective gas or in a high vacuum. In air or in contact with water, it changes into hydrates while absorbing moisture . The compound has a crystal structure of the strontium bromide type with the space group P 4 / nmm (space group no. 129) . At high pressure and 400 ° C, the compound changes into a high pressure variant with an orthorhombic crystal structure and the space group Pbnm (no. 62, position 3) isotypic to that of lead (II) chloride .
Individual evidence
- ↑ a b c d e f g h Georg Brauer (Ed.), With the collaboration of Marianne Baudler u. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6 , p. 1081.
- ↑ a b c Datasheet Europium (II) bromide, 99.99% trace metals basis from Sigma-Aldrich , accessed on May 3, 2014 ( PDF ).
- ↑ E. Yu Tonkov: High Pressure Phase Transformations Handbook 1 . CRC Press, 1992, ISBN 2-88124-758-X , pp. 125 ( limited preview in Google Book Search).



