Fentin acetate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
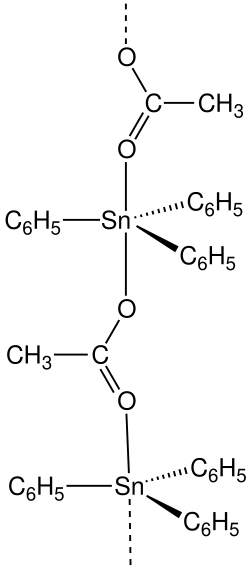
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Fentin acetate | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 20 H 18 O 2 Sn | |||||||||||||||
| Brief description |
colorless solid with an aromatic, slightly acidic odor |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 409.07 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
1.55 g cm −3 |
|||||||||||||||
| Melting point |
122-123 ° C |
|||||||||||||||
| solubility |
|
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Fentin acetate is a chemical compound from the group of organotin compounds and the acetic acid esters of fentin .
Extraction and presentation
Fentin acetate can be obtained by reacting chlorobenzene with magnesium , tin tetrachloride in a Grignard reaction and subsequent reaction with acetic acid.
properties
Fentin acetate is a flammable colorless solid with an aromatic, slightly acidic odor that is practically insoluble in water. It decomposes when heated. Fentin acetate is stable when dry, but hydrolyzes rapidly in the presence of water.
use
Fentin acetate is used as a fungicide . It can also be used as a herbicide against algae in rice fields and as a molluscicide. It was developed by Van der Kerk and Luijten in the 1950s.
Admission
Fentin acetate was approved in the FRG between 1971 and 1997, in the GDR until 1994.
The EU Commission decided in 2002 not to include fentin acetate in the list of permitted active ingredients in pesticides according to Annex 1 of Directive 91/414 / EEC.
In Germany, Austria and Switzerland, no pesticides with this active ingredient are permitted.
Individual evidence
- ↑ a b c d e f g h i Entry on fentin acetate in the GESTIS substance database of the IFA , accessed on January 8, 2020(JavaScript required) .
- ^ Joint Meeting on Pesticide Residues (JMPR), Monograph for Fentin Compounds , accessed December 9, 2014.
- ↑ Entry on Fentin acetate in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on August 1, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ a b Thomas A. Unger: Pesticide Synthesis Handbook . William Andrew, 1996, ISBN 0-8155-1853-6 , pp. 401 ( limited preview in Google Book search).
- ^ Terence Robert Roberts, Terence Robert Roberts DH Hutson: Metabolic Pathways of Agrochemicals: Part 2: Insecticides and Fungicides . Royal Society of Chemistry, 1999, ISBN 0-85404-499-X , pp. 1416 ( limited preview in Google Book search).
- ↑ Entry on fentin acetate at ChemicalBook , accessed January 20, 2014.
- ↑ György Matolcsy, Miklós Nádasy, Viktor Andriska: Pesticide Chemistry . Elsevier, 1988, ISBN 0-444-98903-X , pp. 298 ( limited preview in Google Book search).
- ↑ Peter Brandt: Reports on Plant Protection Products 2009: Active Ingredients in Plant Protection Products ... Springer DE, 2010, ISBN 3-0348-0028-2 , p. 17 ( limited preview in Google Book search).
- ↑ Decision of the Commission of June 20, 2002 on the non-inclusion of fentin acetate in Annex I of Council Directive 91/414 / EEC and the revocation of the authorizations for plant protection products containing this active substance (PDF) , K (2002) 2199.
- ^ Directorate-General for Health and Food Safety of the European Commission: Entry on Fentin acetate in the EU pesticide database ; Entry in the national registers of plant protection products in Switzerland , Austria and Germany ; accessed on February 24, 2016.




