Oryzalin
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
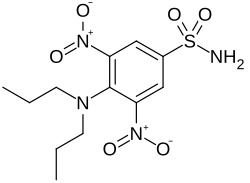
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Oryzalin | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 12 H 18 N 4 O 6 S | |||||||||||||||
| Brief description |
light orange solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 346.36 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
141-142 ° C |
|||||||||||||||
| boiling point |
265 ° C at 760 mmHg |
|||||||||||||||
| Vapor pressure |
<1.3 · 10 −3 mPa (25 ° C) |
|||||||||||||||
| solubility |
|
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Oryzalin is an active ingredient for crop protection and a chemical compound from the group of dinitroanilines .
Extraction and presentation
Oryzalin can be obtained from dipropylamine . The complete synthesis begins with benzene , which is made to react in a multi-stage reaction with chlorine , sulfuric acid , nitric acid , phosphorus oxychloride , ammonia and dipropylamine.
properties
Oryzalin is a light orange, odorless solid that is practically insoluble in water.
use
Oryzalin has been used as a herbicide in berry and viticulture since it was first approved in the USA in 1974 . It is an active ingredient from the group of 2,6-dinitroaniline herbicides and, among other things, inhibits cell division.
Admission
In Switzerland, a plant protection product ( Surflan ) with this active ingredient is approved for combating millet , in Germany and Austria none. Oryzalin has been an approved active ingredient in the states of the European Union since 2011. Currently (2014) pesticide products with oryzalin are approved in Spain and France.
safety instructions
Oryzalin is not poisonous. In studies with rats, however, an increase in the incidence of breast tumors in females and skin and thyroid tumors in both sexes was found.
Web links
- EXTOXNET: Oryzalin
Individual evidence
- ↑ a b c d e data sheet Oryzalin, PESTANAL at Sigma-Aldrich , accessed on May 9, 2017 ( PDF ).
- ↑ a b c d e f EPA: Oryzalin (PDF; 996 kB)
- ↑ a b Terence Robert Roberts, DH Hutson: Metabolic pathways of agrochemicals, Volume 2 . Royal Soc of Chemistry, 1999, ISBN 978-0-85404-499-3 , pp. 265 (English, limited preview in Google Book search).
- ↑ a b Safety data sheet according to 1907/2006 / EG, Article 31. (PDF) In: LGC Standards GmbH. February 9, 2016, accessed July 21, 2020 .
- ^ BASF: Dipropylamine
- ↑ Thomas A. Unger: Pesticide synthesis handbook . 1996, ISBN 978-0-8155-1401-5 , pp. 871 (English, limited preview in Google Book search).
- ^ Entry on Oryzalin in the Hazardous Substances Data Bank , accessed July 27, 2012.
- ↑ a b Directorate-General for Health and Food Safety of the European Commission: Entry on oryzalin in the EU pesticide database; Entry in the national registers of plant protection products in Switzerland , Austria and Germany ; accessed on March 13, 2016.
- ↑ Directive 2011/27 / EU of the Commission of March 4, 2011 amending Council Directive 91/414 / EEC to include the active substance oryzalin and amending Commission Decision 2008/934 / EC In: Official Journal of the European Union . L 60, March 5, 2011, pp. 12-16.


