Osmium (IV) oxide
| Crystal structure | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
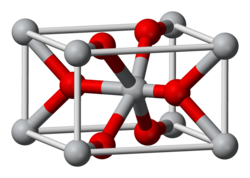
|
|||||||||||||
| __ Os 4+ __ O 2− | |||||||||||||
| General | |||||||||||||
| Surname | Osmium (IV) oxide | ||||||||||||
| other names |
Osmium dioxide |
||||||||||||
| Ratio formula | OsO 2 | ||||||||||||
| Brief description |
|
||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 222.20 g mol −1 | ||||||||||||
| Physical state |
firmly |
||||||||||||
| density |
11.4 g cm −3 |
||||||||||||
| Melting point |
650 ° C (decomposition) |
||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Osmium (IV) oxide is an inorganic chemical compound of osmium from the group of oxides .
Extraction and presentation
Osmium (IV) oxide can be obtained by reducing osmium tetroxide in a hydrogen - nitrogen gas stream or by reacting potassium hexachloro- osmate K 2 OsCl 6 with sodium carbonate at high temperatures.
Single crystals of the oxide can be produced using a CVT (Chemical Vapor Transport) method . For this purpose, elemental osmium is first converted to the polycrystalline oxide with sodium chlorate in a melted and evacuated ampoule in a 2-stage reaction at initially 300 ° C and then 650 ° C. The ampoule with the oxide is then placed in a 2-zone oven so that half of the ampoule is in one of the zones. While one half of the ampoule is heated to 960 ° C, the other half is kept at 900 ° C. After a reaction time of 2 days (with 0.15 g of elemental osmium initially used), the ampoule is allowed to cool and, after opening, golden single crystals of the target compound about 2 mm in size are obtained.
properties
Osmium (IV) oxide is purchased as a black solid that is insoluble in water, while the freshly made compound is golden in color in both poly- and monocrystalline forms. In air it slowly changes into osmium tetroxide and can react pyrophorically with atmospheric oxygen. It has a crystal structure of rutile type (a = 451 microns, c = 319 pm). The dihydrate is a deep black powder which, like the anhydrate, tends to form osmium tetroxide in air. It is soluble in hydrochloric acid to form dihydrogen hexachloro-osmate H 2 [OsCl 6 ].
Individual evidence
- ↑ a b c d e f data sheet Osmium (IV) oxide, Os 83% min at AlfaAesar, accessed on September 1, 2013 ( PDF )(JavaScript required) .
- ↑ a b c D. B. Rogers et al .: Single crystals of transition-metal dioxides - B. Osmium dioxide . In: FA Cotton (Ed.): Inorganic Syntheses . tape 13 . McGraw-Hill Book Company, Inc., 1972, ISBN 07-013208-9 ( defective ) , p. 135-145 (English).
- ^ Jean d'Ans, Ellen Lax, Roger Blachnik: Pocket book for chemists and physicists . Springer DE, 1998, ISBN 3-642-58842-5 , pp. 646 f . ( limited preview in Google Book search).
- ↑ a b Georg Brauer (Ed.) U. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume III, Ferdinand Enke, Stuttgart 1981, ISBN 3-432-87823-0 , p. 1745.


