Phenylarsine dichloride
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
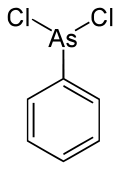
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Phenyldichlorarsine | |||||||||||||||
| Molecular formula | C 6 H 5 AsCl 2 | |||||||||||||||
| Brief description |
colorless, unpleasant smelling liquid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 222.93 g · mol -1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
1.66 g cm −3 |
|||||||||||||||
| Melting point |
−15.6 ° C |
|||||||||||||||
| boiling point |
255 ° C |
|||||||||||||||
| Vapor pressure |
0.15 h Pa (at 20 ° C) |
|||||||||||||||
| Refractive index |
1.6371 |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
Phenylarsine dichloride , also known as dichlorophenylarsine, was first used as a chemical warfare agent by German armies as "Pfiffikus" in October 1917. The French call it “Sternite”, the English call it “DJ” and the Americans “PD”.
History and manufacture
It was first made in 1878 by La Coste and Michaelis by direct synthesis from benzene and arsenic trichloride . It was used by the German Wehrmacht as the main component of so-called arsine oil for mixing with yperite . The reaction of phenyldichloroarsine with diphenylamine , in which 10-chloro-5,10-dihydrophenarsazine ( adamsite ) is formed, is interesting .
Chemical properties
In its pure form it is a colorless liquid with a boiling point of 256 ° C and a fixed point of −20 ° C. The compound is insoluble in water and there is no noticeable hydrolysis even when heated. Chemically, phenylarsine dichloride behaves analogously to Lewisite .
Poisonous effect
It has a strong irritant effect on the upper respiratory tract. As an inhalation poison, it is damaging to the lungs; inhalation leads to bronchopneumonia and pulmonary edema . Depending on the inhaled dose, the poisoning can also be fatal. The skin-damaging effect is not as strong as with Lewisite, it works like this, but without a latency period. It forms blisters with a relatively good tendency to heal. The damage to the eyes is more severe than that of sulfur leyperites, but has relatively better healing tendencies.
The reason for the relatively strong poisonous effect is the trivalent arsenic atom. Trivalent arsenic compounds are always superior to the pentavalent arsenic compounds in terms of their toxic effects.
Individual evidence
- ↑ a b c d e f Entry on dichlorophenylarsine in the GESTIS substance database of the IFA , accessed on February 1, 2016(JavaScript required) .
- ↑ Yambushev, FD; Kokorev, GI; Khalitov, FG; Tenisheva, N. Kh .; Kut'in, S. V: Synthesis and properties of arylarsonous dichlorides . In: Journal of general chemistry of the USSR . tape 53 , no. 12 , 1983, ISSN 0022-1279 , pp. 2718-2722 .
- ↑ Not explicitly listed in Regulation (EC) No. 1272/2008 (CLP) , but with the specified labeling it falls under the group entry arsenic compounds, with the exception of those named in this appendix in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA) on February 1, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ Landolt-Börnstein Online Search: European regulations regarding dichlorophenylarsine
literature
- Haas, R. et al. (1998): Chemical-analytical investigation of arsenic warfare agents and their metabolites. UWSF - Z Umweltchem Ecotox ; 10, pp. 289-293; PDF (free full text access)

