Pimecrolimus
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
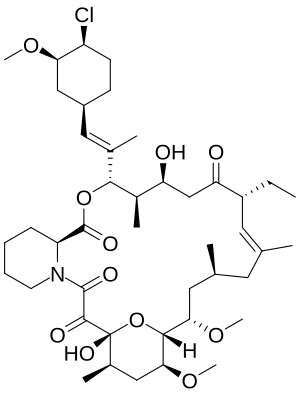
|
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Non-proprietary name | Pimecrolimus | |||||||||||||||||||||
| other names |
|
|||||||||||||||||||||
| Molecular formula | C 43 H 68 ClNO 11 | |||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Drug information | ||||||||||||||||||||||
| ATC code | ||||||||||||||||||||||
| Drug class | ||||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 810.45 g · mol -1 | |||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Pimecrolimus ( trade name Elidel , manufacturer: MEDA) is a medicinal substance (approval in EU: October 2002) that is used to treat atopic eczema ("neurodermatitis"). As such, it belongs to the group of topical immunomodulators , which also includes tacrolimus .
structure
It is a very lipophilic compound of the macrolide type . The structure was found in the ferment of an Ascomycete species (hose fungus).
Mechanism of action
Similar to the mechanism of action of cyclosporin , pimecrolimus binds intracellularly to an immunophilin (macrophilin-12). This inhibits calcineurin and consequently the activity of the T cells is reduced. These now synthesize fewer cytokines and other inflammatory mediators . The release of inflammatory mediators from mast cells is also suppressed, which leads to a reduced immune response .
use
Pimecrolimus is used as a cream to apply to the affected skin in various forms of atopic eczema . It is used for both short-term and long-term treatment, in which case interval therapy is recommended. Because they are better tolerated than with glucocorticoids, the possibility of extensive treatment and prophylactic use is given. It is now being used increasingly for rosacea and seborrheic eczema , also as a replacement for glucocorticoids. As with the treatment of lichen sclerosus , this is an off-label use . The active ingredient is also used as an off-label use for the treatment of balanitis circinata in order to completely suppress the visible consequences of this rheumatic disease.
Side effects
Skin reactions in the form of burning, redness and itching are very common, especially at the beginning of treatment. There is also an increased risk of bacterial infections and viral diseases due to the immunosuppressive effect. An increased risk of tumors was initially discussed, which is why studies to clarify this risk were commissioned in the USA and Europe. A study published in February 2015 with 7457 children (26792 person-years) found no evidence of an increased risk of cancer with topical application.
For the time being, the health authorities issued recommendations on the use of topical immunomodulators, which include restrictions for children up to 2 years of age and for people with weakened immune systems.
Trade names
Elidel (D, A); several reimports
literature
- JG Meingassner, M. Grassberger, H. Fahrngruber, HD Moore, H. Schuurman, A. Stütz: A novel anti-inflammatory drug, SDZ ASM 981, for the topical and oral treatment of skin diseases: in vivo pharmacology . In: Br J Dermatol , 1997, 137 (4), pp. 568-576. PMID 9390333 .
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Success with pimecrolimus in eczema and rosacea . In: ÄrzteZeitung , May 10, 2005; accessed on May 12, 2014.
- ↑ Topical pimecrolimus is an effective treatment for balanitis circinata erosiva . PMID 16207202
- ↑ FDA Health Warning about Pimecrolimus, March 10, 2005 ( Memento of May 6, 2008 in the Internet Archive )
- ↑ David J. Margolis, Katrina Abuabara, Ole J. Hoffstad, Joy Wan, Denise Raimondo: Association Between Malignancy and Topical Use of Pimecrolimus . In: JAMA dermatology . tape 151 , no. 6 , June 1, 2015, p. 594-599 , doi : 10.1001 / jamadermatol.2014.4305 , PMID 25692459 , PMC 4465068 (free full text).