Resazurin
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Resazurin | ||||||||||||||||||
| other names |
7-Hydroxy-3 H -phenoxazin-3-one-10-oxide |
||||||||||||||||||
| Molecular formula | C 12 H 7 NO 4 | ||||||||||||||||||
| Brief description |
Dark red, narrow crystals with a greenish sheen |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 229.18 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
> 250 ° C |
||||||||||||||||||
| boiling point |
decomposition |
||||||||||||||||||
| pK s value |
6.71 |
||||||||||||||||||
| solubility |
|
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
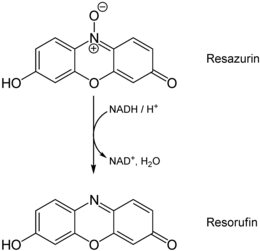
Resazurin (synonym: Alamar blue ) is a dye that is used in chemistry as a redox indicator and pH indicator . Resazurin can display both redox reactions and changes in pH . It is used as a reagent for hyposulfites and to measure cell viability and the cytotoxicity of substances. It can be made from resorcinol .
properties
Resazurin is a non-toxic, water-soluble blue redox -dye, the at reduction irreversibly to pink, fluorescent resorufin is implemented. In a second reversible reaction, resorufin is reduced to colorless dihydroresorufin.
In an acidic aqueous solution, the indicator has an orange color that changes to dark purple from a pH value of 6.5.
Resazurin is mainly marketed as the sodium salt. The light-sensitive substance can be stored as a solution at temperatures between 2 and 8 ° C for up to a year, the salt can be stored dry at room temperature.
Color of the aqueous solution
An aqueous Resazurin solution appears blue in thin layers or diluted concentrations; thicker layers and higher concentrations show a red color. This material property is called dichromatism or polychromatism .
use
Resazurin is used in current medical-chemical research as a measure of cell viability to measure the cytotoxicity of substances. The Cytotox Assay, or Alamar Blue Assay, uses an aqueous resazurin solution that gradually reduces normally working cells to resorufin. Cytotoxic substances lower the metabolic activity and the redox potential of the cell , which, depending on the toxic effectiveness, slows down or stops the reduction of the resazurin.
The red bottle experiment uses resazurin as a redox indicator. In this case, glucose in a basic solution reversibly reduces the resorufin formed from the resazurin to a colorless form, which is oxidized again to the pink form by the oxygen in the air by shaking in an open vessel. This process can be repeated over a period of several hours.
toxicology
Resazurin is classified as non-toxic, but like many chemicals, it can cause health problems if swallowed. In addition, the indicator irritates eyes and skin on contact.
Individual evidence
- ↑ a b c d e f g Ram Wasudeo Sabnis: Handbook of acid-base indicators. CRC Press, 2007, ISBN 978-0-8493-8218-5 , p. 337.
- ↑ a b Resazurin sodium salt data sheet from Sigma-Aldrich , accessed on April 22, 2011 ( PDF ).
- ↑ a b R. S. Twigg: Oxidation-Reduction Aspects of Resazurin. In: Nature. 155, No. 3935, 1945, pp. 401-402, doi : 10.1038 / 155401a0 .
- ↑ S. Kreft, M. Kreft: Quantification of dichromatism - a characteristic of color in transparent materials. In: Journal of the Optical Society of America A: Optics, Image Science & Vision. 26, 2009, pp. 1576-1581. doi : 10.1364 / JOSAA.26.001576 .
- ↑ Resazurin data sheet at AlfaAesar, accessed on March 23, 2010 ( PDF )(JavaScript required) .
Web links
- R&D Systems (Ed.): Resazurin. (Properties and use, PDF file 51 kB, English)
- Paul G. Tratnyek et al .: Visualizing Redox Chemistry: Probing Environmental Oxidation-Reduction Reactions with Indicator Dyes. In: The Chemical Educator. 6, No. 3, 2001, pp. 172-179, doi : 10.1333 / s00897010471a (treatise on selected redox dyes, English)
- Blue Bottle, Red and Blue Bottle (To the Red Bottle attempt, English)
