Rubidium oxalate
| Structural formula | |||||||
|---|---|---|---|---|---|---|---|
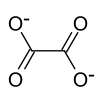
|
|||||||
| General | |||||||
| Surname | Rubidium oxalate | ||||||
| Molecular formula | Rb 2 C 2 O 4 | ||||||
| Brief description |
colorless, dull crystals |
||||||
| External identifiers / databases | |||||||
|
|||||||
| properties | |||||||
| Molar mass | |||||||
| Physical state |
firmly |
||||||
| density |
2.76 g cm −3 (monohydrate) |
||||||
| Melting point |
decomposition |
||||||
| safety instructions | |||||||
|
|||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||
Rubidium oxalate is the rubidium salt of oxalic acid .
Manufacturing
Rubidium oxalate can be made from rubidium carbonate and oxalic acid.
It is also formed during the thermal decomposition of rubidium formate .
properties
Rubidium oxalate crystallizes as monohydrate (COO) 2 Rb 2 in the monoclinic crystal system. and is isomorphic to potassium oxalate monohydrate. There are two modifications of the anhydrate at room temperature : one modification is monoclinic and isotypic to cesium oxalate , the other is orthorhombic and isotypic to potassium oxalate. Freshly produced anhydrous rubidium oxalate initially mainly contains the monoclinic phase , but this slowly converts irreversibly into the orthorhombic modification. In 2004, two more high temperature phases of rubidium oxalate were discovered. Crystal data of the various modifications of rubidium oxalate:
| modification | Crystal system | Space group | a in Å | b in Å | c in Å | β | Z |
|---|---|---|---|---|---|---|---|
| alpha | monoclinic | P2 1 / c | 6.328 | 10.455 | 8.217 | 98.016 ° | 4th |
| beta | orthorhombic | Pbam | 11.288 | 6.295 | 3,622 | - | 2 |
| Monohydrate | monoclinic | C2 / c | 9.617 | 6.353 | 11.010 | 109.46 ° | 4th |
The standard enthalpy of formation of the crystalline rubidium oxalate is 1325.0 ± 8.1 kJ / mol.
The decomposition of rubidium oxalate with the release of carbon monoxide and subsequently carbon dioxide and oxygen takes place at 507 - 527 ° C.
In addition to the neutral rubidium oxalate, there is also a hydrogen oxalate with the formula RbH (COO) 2 , which is isomorphic to the corresponding potassium compound and forms monoclinic crystals, as well as an acidic tetraoxalate with the formula RbH 3 (COO) 4 , which crystallizes as a dihydrate, at 18 ° C has a density of 2.125 g / cm −3 and a solubility of 21 g / l at 21 ° C.
When a solution in hydrogen peroxide is evaporated, rubidium oxalate forms a monoperhydrate with the composition (COO) 2 Rb 2 · H 2 O 2 , which forms monoclinic crystals that are relatively stable in air.
Rubidium oxalate reacts with hydrogen fluoride to form a complex compound.
Individual evidence
- ^ A b c Jean D'Ans, Ellen Lax: Pocket book for chemists and physicists. 3. Elements, inorganic compounds and materials, minerals, volume 3. 4th edition, Springer, 1997, ISBN 978-3-540-60035-0 , pp. 686f. ( limited preview in Google Book search)
- ↑ a b c Dissertation: "Conformational clarification of inorganic oxo anions of carbon", Sascha Vensky, University of Stuttgart, 2004. S. 117ff. PDF
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ^ E. Giglio, S. Loreti, NV Pavel: "EXAFS: A New Approach to the Structure of Micellar Aggregates" in J. Phys. Chem. , 1988 , 92 , pp. 2858-2862. doi: 10.1021 / j100321a032
- ^ A b T. Meisel, Z. Halmos, K. Seybold, E. Pungor: "The thermal decomposition of alkali metal formats" in Journal of Thermal Analysis and Calorimetry 1975 , 7 (1). Pp. 73-80. doi: 10.1007 / BF01911627
- ↑ Björn Pedersen: “The Equilibrium Hydrogen-Hydrogen Distances in the Water Molecules in Potassium and Rubidium Oxalate Monohydrates” in Acta Cryst. , 1966 , 20 , p. 412ff. doi: 10.1107 / S0365110X66000951
- ^ A b c R. E. Dinnebier, S. Vensky, M. Panthöfer, M. Jansen: "Crystal and molecular structures of alkali oxalates: first proof of a staggered oxalate anion in the solid state." in Inorg. Chem , 2003 , 42 (5), pp. 1499-1507. PMID 12611516 .
- ↑ Robert E. Dinnebier, Sascha Vensky, Martin Jansen, Jonathan C. Hanson: Crystal Structures and Topological Aspects of the High ‐ Temperature Phases and Decomposition Products of the Alkali ‐ Metal Oxalates M 2 [C 2 O 4 ] (M = K, Rb, Cs) . In: Chemistry - A European Journal . tape 11 , no. 4 , February 4, 2005, p. 1119-1129 , doi : 10.1002 / chem.200400616 .
- ↑ Takuya Echigo, Mitsuyoshi Kimata: The common role of water molecule and lone electron pair as a bond-valence mediator in oxalate complexes: the crystal structures of Rb 2 (C 2 O 4 ) H 2 O and Tl 2 (C 2 O 4 ) . In: Journal of Crystallography . tape 221 , no. November 12 , 2006, pp. 762–769 , doi : 10.1524 / zkri.2006.221.12.762 .
- ↑ Y. Masuda, H. Miyamoto, Y. Kaneko, K. Hirosawa: "The standard molar enthalpies of formation of crystalline rubidium and cesium oxalates" in J. Chem. Thermodynamics , 1985 , 17 (2), pp. 159-164 . doi: 10.1016 / 0021-9614 (85) 90068-0
- ↑ J. Piccard: "Contribution to the knowledge of the rubidium compounds " in Journal für Praktische Chemie 1862 , 86 (1), pp. 449-460. doi: 10.1002 / prac.18620860163 full text
- ^ H. Watts: "A dictionary of chemistry and the allied branches of other sciences", Volume 4, Verlag Longmans, Green and Co., 1866, p. 264. ( limited preview in the Google book search)
- ^ R. Abegg, F. Auerbach: "Handbuch der inorganic Chemie". Verlag S. Hirzel, Vol. 2, 1908. P. 435. Full text
- ↑ BF Pedersen: "The Crystal Structure of Potassium and Rubidium Oxalate Monoperhydrates, K 2 C 2 O 4 .H 2 O 2 and Rb 2 C 2 O 4 .H 2 O 2 " in Acta Chem. Scand. 1967 , 21 , pp. 779-790. doi: 10.3891 / acta.chem.scand.21-0779
- ↑ RF Weinland, W. Stille: "About the addition of crystalline hydrogen fluoride to oxalates and ammonium tartrate" in Justus Liebigs Annalen der Chemie 1903 , 328 (2), pp. 149-153. doi: 10.1002 / jlac.19033280205





