Boil

Boil , also referred to as cooking and Wallen , is, in contrast to evaporation , rapid evaporation , so a faster phase transition from cash to Gaseous where the, vapor pressure of a liquid to the ambient pressure reached. Both pure substances and mixtures can boil .
Boiling process
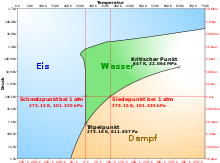
If the saturation vapor pressure of a liquid corresponds to the total pressure (the sum of the partial pressures ) or exceeds this, the boiling point has been reached and the substance boils.
As long as the pressure remains constant, when pure substances (e.g. water ) boil , the temperature also remains constant until the liquid has completely evaporated. All the heat supplied is invested in the phase transition . This is the basis of the very effective evaporative cooling of objects with high thermal loads.
In the case of mixtures, for example with the components water and acetone , the composition of the liquid and gas phases is usually different, the higher-boiling component accumulates in the liquid phase. Therefore, the boiling point increases as evaporation proceeds. The boiling process is best described by a boiling diagram . In this case the components are present in the liquid and the gaseous phase in different concentrations. This fact is used in thermal separation processes such as distillation .
Example water
Under normal conditions, i.e. at an air pressure of 1013 hPa and a temperature of 20 ° C, water is liquid. If water in an open-topped vessel is heated from below by a heat source , a temperature gradient builds up . It is hottest on the ground, the surface temperature is still the same as the air temperature . Since warm water has a lower density than cold water, it rises, but colder water sinks down ( convection ). If heated slowly, all of the water can evaporate without bubbles rising.
If the temperature of the soil is higher than the temperature of the boiling point at the prevailing hydrostatic pressure, the water evaporates. First, bubbles form on small unevenness in the floor ( nucleation ) , which rise up and allow cooler water to flow down: the water begins to boil. The bubbles contain water vapor or other gases that were dissolved in the cold water.
As long as bubbles rise and an intensive heat transfer through the wall is guaranteed, one speaks of nucleate boiling . If, on the other hand, a coherent vapor layer forms, this is known as film boiling .
During the transition from nucleate boiling to film boiling, the heat transfer between soil and water deteriorates suddenly. With nucleate boiling the water is stirred by the rising bubbles, this is missing with film boiling. This effect must be taken into account when cooling hot objects (such as fuel assemblies ) with water.
At steady boiling , the boiling temperature has not yet been reached in the entire vessel. The bubbles condense in the colder water and collapse. The upper layers of the water are additionally heated by the rising hot steam bubbles until the entire body of water is evenly heated.
The warming of the surface water also leads to a warming of the vapor phase above. If the surface temperature of the water is higher than the boiling point at the prevailing air pressure (e.g. higher than 100 ° C at 1013 hPa), the water evaporates immediately at the rate at which the heat required for evaporation is supplied.
Delayed boiling
Water, hydrogen peroxide or alkalis ( e.g. caustic soda ) without dust particles or gas bubbles can also be heated above the boiling point in clean vessels without boiling. The smallest disturbances, such as vibrations that result in thorough mixing, can lead to an explosive separation of the liquid from the vapor phase, which is known as boiling delay . To avoid this, solutions are added with boiling stones made of clay or pumice stone , which are not attacked by the solution, but which facilitate the formation of small bubbles due to their porous structure so that there is no delay in boiling.
Physical explosion
The strong increase in volume of a substance due to evaporation can lead to a physical explosion if a lot of heat is suddenly applied .
See also
- Simmering (cooking)
- Saturation vapor pressure
- Leidenfrost effect
- Evaporative cooling
- Leimsieder · unlucky boilers · Saltworks · Seifensieder
Individual evidence
- ↑ a b Ernst Doering, Herbert Schedwill, Martin Dehli: Fundamentals of technical thermodynamics . 2008, ISBN 978-3-8351-0149-4 ( page 96 in the Google book search).
- ↑ Claus Czeslik, Heiko Seemann, Roland Winter: Basic knowledge of physical chemistry . 2007, ISBN 978-3-8351-0047-3 ( page 104 in the Google book search).