Tin (IV) fluoride
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| __ Sn 4+ __ F - | ||||||||||||||||
| Crystal system |
tetragonal |
|||||||||||||||
| Space group |
I 4 / mmm (No. 139) |
|||||||||||||||
| General | ||||||||||||||||
| Surname | Tin (IV) fluoride | |||||||||||||||
| other names |
Tin tetrafluoride |
|||||||||||||||
| Ratio formula | SnF 4 | |||||||||||||||
| Brief description |
colorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 194.70 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
4.78 g cm −3 |
|||||||||||||||
| Melting point |
705 ° C (sublimation) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Tin (IV) fluoride is an inorganic chemical compound of tin from the group of fluorides .
Extraction and presentation
Tin (IV) fluoride can be obtained by reacting tin (IV) chloride with hydrogen fluoride .
properties
Tin (IV) fluoride is a colorless, extremely hygroscopic solid that dissolves in water with strong hissing.
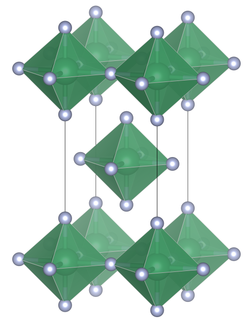
Like lead (IV) fluoride, the connection consists of SnF 6 octahedra which are linked to planar layers via common equatorial bridges with four SnF 6 octahedra each .
use
Tin (IV) fluoride is used as an additive to prevent tooth decay in toothpaste.
Individual evidence
- ↑ a b c d e data sheet Tin (IV) fluoride from Sigma-Aldrich , accessed on May 25, 2017 ( PDF ).
- ↑ a b c d Georg Brauer (Ed.), With the collaboration of Marianne Baudler a . a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6 , p. 232.
- ^ AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 101st edition. Walter de Gruyter, Berlin 1995, ISBN 3-11-012641-9 , p. 970.
- ^ Catherine E. Housecroft, Edwin C. Constable: Chemistry: An Introduction to Organic, Inorganic and Physical Chemistry . Pearson Education, 2010, ISBN 0-273-73308-7 , pp. 769 ( limited preview in Google Book search).

