3,4-hexanedione
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
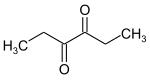
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | 3,4-hexanedione | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 6 H 10 O 2 | ||||||||||||||||||
| Brief description |
yellow liquid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 114.14 g mol −1 | ||||||||||||||||||
| Physical state |
liquid |
||||||||||||||||||
| density |
0.939 g cm −3 |
||||||||||||||||||
| Melting point |
−10 ° C |
||||||||||||||||||
| boiling point |
131 ° C |
||||||||||||||||||
| Vapor pressure |
9.9 hPa (20 ° C) |
||||||||||||||||||
| solubility |
|
||||||||||||||||||
| Refractive index |
1.41 (20 ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | |||||||||||||||||||
3,4-Hexanedione is a chemical compound from the group of alkanediones .
Occurrence
3,4-hexanedione is found naturally in cauliflower , honey, and coffee .
Extraction and presentation
3,4-Hexanedione can be obtained by rearrangement of 3,4-dihydroxy-1,5-diene with a ruthenium catalyst. It can also be prepared by condensation of ethyl propionate in the presence of sodium metal, followed by oxidation of the propionine obtained with copper acetate or iron (III) chloride .
properties
3,4-hexanedione is a yellow liquid that is easily soluble in water. Your aqueous solution is acidic. The compound has an aromatic, toasted, burnt, buttery, nutty, caramel-like, pungent odor.
use
3,4-Hexanedione is being investigated as a substitute for diacetyl for butter flavors. It is also used as a flavoring agent.
safety instructions
The vapors of 3,4-hexanedione can form an explosive mixture with air ( flash point 27 ° C, ignition temperature 250 ° C).
Individual evidence
- ↑ a b c d e f g h i j k Entry on 3,4-hexanedione in the GESTIS substance database of the IFA , accessed on June 12, 2019(JavaScript required) .
- ↑ a b c d e f George A. Burdock: Fenaroli's Handbook of Flavor Ingredients . CRC Press, 2004, ISBN 978-1-4200-3787-6 , pp. 1947 ( limited preview in Google Book Search).
- ↑ a b c data sheet 3,4-Hexanedione, ≥95%, FG from Sigma-Aldrich , accessed on June 12, 2019 ( PDF ).
- ↑ Houben-Weyl Methods of Organic Chemistry Vol. E 18, 4th Edition Supplement Organo -? - Metal Compounds as Auxiliaries in Organic Chemistry . Georg Thieme Verlag, 2014, ISBN 3-13-182004-7 , p. 625 ( limited preview in Google Book search).
