Americium (III) oxide
| Crystal structure | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
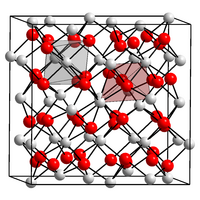
|
||||||||||
| __ On 3+ __ O 2− | ||||||||||
| Space group |
Ia 3 (No. 206) |
|||||||||
| Lattice parameters |
a = 1103 pm |
|||||||||
| General | ||||||||||
| Surname | Americium (III) oxide | |||||||||
| other names |
|
|||||||||
| Ratio formula | On 2 O 3 | |||||||||
| Brief description |
red-brown solid |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 534.121 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| Melting point |
2205 ° C |
|||||||||
| Hazard and safety information | ||||||||||
 Radioactive |
||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Americium (III) oxide is an oxide of the element americium . It has the molecular formula Am 2 O 3 . Since all isotopes of americium are only artificially produced, americium (III) oxide does not occur naturally.
presentation
Americium (III) oxide can be obtained by thermal decomposition of americium (IV) oxide in a vacuum :
Another way is given by the reduction of americium (IV) oxide with molecular hydrogen :
properties
Americium (III) oxide is a red-brown solid and has a melting point of 2205 ° C. Three modifications are known. The α-form crystallizes in the hexagonal crystal system in the space group P 3 m 1 (space group no. 156) and the lattice parameters a = 380.5 pm and c = 596 pm. The β-form of americium (III) oxide has a monoclinic crystal system with the lattice parameters a = 1438 pm, b = 352 pm, c = 892 pm. The γ-americium (III) oxide crystallizing in γ- manganese (III) oxide form has the lattice parameter a = 1103 pm and the space group Ia 3 (No. 206) .
safety instructions
Classifications according to the CLP regulation are not available because they only include chemical hazard and play a completely subordinate role compared to the hazards based on radioactivity . The latter also only applies if the amount of substance involved is relevant.
Individual evidence
- ^ A b c A. F. Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 , p. 1972.
- ↑ The hazards emanating from radioactivity do not belong to the properties to be classified according to the GHS labeling. With regard to other hazards, this substance has either not yet been classified or a reliable and citable source has not yet been found.
- ↑ a b Gmelin's Handbook of Inorganic Chemistry , System No. 71, Transurane, Part C, p. 33.
- ↑ a b T. D. Chikalla, L. Eyring: Phase Relationships in the Americium Oxygen System , in: J. Inorg. Nucl. Chem. , 1968 , 30 (1), pp. 133-145 ( doi : 10.1016 / 0022-1902 (68) 80072-7 ).
- ^ DH Templeton, CH Dauben: Crystal Structures of Americium Compounds , in: J. Am. Chem. Soc. , 1953 , 75 (18), pp. 4560-4562 ( doi : 10.1021 / ja01114a051 ).
literature
- Wolfgang H. Runde, Wallace W. Schulz: Americium , in: Lester R. Morss, Norman M. Edelstein, Jean Fuger (Eds.): The Chemistry of the Actinide and Transactinide Elements , Springer, Dordrecht 2006; ISBN 1-4020-3555-1 , pp. 1265-1395 ( doi : 10.1007 / 1-4020-3598-5_8 ).
![{\ displaystyle {\ ce {4 AmO2 -> [\ Delta T] 2 Am2O3 + O2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/b16fbf9f2ce9a15dbf1314f816e2f5b54883a34e)
