Ammonium iodide
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
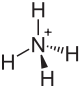 |
||||||||||||||||
| General | ||||||||||||||||
| Surname | Ammonium iodide | |||||||||||||||
| other names |
Iodammonium |
|||||||||||||||
| Molecular formula | NH 4 I. | |||||||||||||||
| Brief description |
white, odorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 144.94 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
2.51 g cm −3 (20 ° C) |
|||||||||||||||
| Melting point | ||||||||||||||||
| solubility |
very light in water (1770 g l −1 at 25 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Ammonium iodide is the ammonium salt of hydrogen iodide .
properties
Ammonium iodide forms white , cubic crystals that are easily soluble in water and ethanol . It is easily decomposable and turns yellow to brown in air as a result of the oxidation of the iodide ion to iodine I 2 . The compound occurs in three crystal structures, the low-temperature form having a tetragonal crystal structure with the space group P 4 / nmm (space group no. 129) .
synthesis
Ammonium iodide can be produced by the reaction of iodine or hydrogen iodide with ammonia or ammonium carbonate :
However, caution should be exercised in the reaction between iodine I 2 and ammonia water, since iodine nitrogen is formed very easily .
In the laboratory, ammonium iodide solution is similarly obtained from 10% ammonia water , iodine and hydrogen peroxide :
use
Ammonium iodide is used in the phototechnical industry and in medicine.
Individual evidence
- ↑ a b c d Entry on ammonium iodide in the GESTIS substance database of the IFA , accessed on January 9, 2019(JavaScript required) .
- ↑ a b Data sheet Ammonium iodide, 98 +% from AlfaAesar, accessed on February 1, 2019 ( PDF )(JavaScript required) .
- ^ Ammonium iodide - article in Meyers Konversationslexikon, Verlag des Bibliographisches Institut, Leipzig and Vienna, fourth edition, 1885-1892
- ^ High Pressure Phase Transformations: A Handbook . CRC Press, 1992, ISBN 978-2-88124-759-0 , pp. 445 ( books.google.de ).
- ↑ G. Brauer (Ed.), Handbook of Preparative Inorganic Chemistry 2nd ed., Vol. 1, Academic Press 1963, pp. 289-90.




