Ammonium fluoride
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
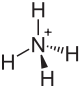 |
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Ammonium fluoride | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | NH 4 F | ||||||||||||||||||
| Brief description |
white, deliquescent crystals |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 37.04 g · mol -1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| density |
1.01 g cm −3 |
||||||||||||||||||
| Melting point |
Decomposition from 100 ° C |
||||||||||||||||||
| solubility |
easily in water (820 g l −1 at 20 ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| MAK |
1 mg m −3 (inhalable aerosol fraction) |
||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Ammonium fluoride is the ammonium salt of hydrogen fluoride .
properties
Ammonium fluoride forms colorless, easily deliquescent, easily soluble in water crystals and is poisonous.
The substance is able to attack glass and should therefore preferably be stored in containers made of polyethylene (PE), polyvinyl chloride (PVC) or glass bottles covered with paraffin .
Reactions
When heated, it breaks down into ammonia and hydrogen fluoride:
synthesis
Ammonium fluoride is produced by neutralizing aqueous ammonia solution with hydrofluoric acid and then crystallizing from water.
Another synthesis route is the heating of ammonium chloride with sodium fluoride , whereby the product sublimates:
use
Ammonium fluoride is used for glass etching and, like hydrofluoric acid , is used in analysis to break down silicates .
Precautions
Ammonium fluoride is poisonous. Avoid contact with skin and eyes. Hydrogen fluoride is released on contact with strong acids. This is extremely poisonous and extremely corrosive.
disposal
Precipitate dissolved fluoride as calcium fluoride .
See also
Web links
Individual evidence
- ↑ Entry on AMMONIUM FLUORIDE in the CosIng database of the EU Commission, accessed on February 12, 2020.
- ↑ a b c d e f g h Entry on ammonium fluoride in the GESTIS substance database of the IFA , accessed on January 10, 2017(JavaScript required) .
- ↑ Entry on ammonium fluoride in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on August 1, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ a b Helmut Hörath: Dangerous substances and preparations . 6th edition. Wissenschaftliche Verlagsgesellschaft mbH, Stuttgart 2002, ISBN 3-8047-1850-7 , p. 410 .
- ↑ G. Brauer (Ed.), Handbook of Preparative Inorganic Chemistry. 2nd ed., Vol. 1, Academic Press 1963, ISBN 012126601X , p. 183




