Base analogues
Base analogs are analogs of the nucleobases and count among the antimetabolites . They can change the genetic material of a cell. The mutagenic effect is based on the alteration of individual nucleobases in the DNA .
Base analogs are chemically similar to the nucleobases of DNA and often differ from them only in one active group . By changing the molecular structure, these bases are able to pair complementarily with different bases, which can cause a point mutation in the DNA. As a result of a point mutation in a gene , the protein formed from it can also have a mutation and is possibly no longer functional.
Base analogues can, but need not, hinder transcription. Here the modification is important, whether it sterically interferes with the DNA-dependent DNA polymerase .
Examples
- 5-bromouracil (5-BU), 5-fluorouracil (5-FU), 5-hydroxymethyluracil or 6-methyluracil are derived from uracil , in which hydrogen is replaced by bromine or fluorine. Through keto-enol tautomerism , 5-BU or 5-FU is able to pair with both adenine (in the keto form ) and with guanine (in the enol form ). This can trigger transitions .
Nucleobase Methyl derivative Fluorine derivative Chlorine derivative Bromine derivative 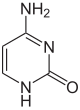
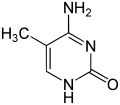


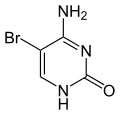
Cytosine 5-methylcytosine 5-fluorocytosine 5-chlorocytosine 5-bromocytosine 


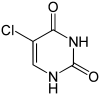

Uracil 5-methyluracil = thymine (!) 5-fluorouracil 5-chloruracil 5-bromouracil
- 2-aminopurine (2-AP) is an isomer of adenine (6-aminopurine). Through imine-enamine tautomerism , 2-AP is able to pair with both thymine (in the amino form ) and with cytosine (in the imino form ). This can trigger transitions .


Adenine 2-aminopurine ( isoadenine )
Individual evidence
- ↑ James D. Watson, Stephen P. Bell, Tania A. Baker, Alexander Gann, Michael Levine, Richard Losick: Molecular Biology . Pearson Studies - Biology 2011. ISBN 9783868940299 . P. 307f.