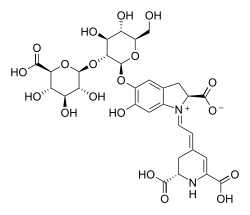
Betalaine

Betalaines are a group of chemically very similar water-soluble flower and fruit dyes , in addition to anthocyanins and carotenoids form the third group of vegetable dyes. They occur exclusively in clove-like plants (with the exception of the clove family and Molluginaceae ) and leaf mushrooms . Pharmacologically, they are non-toxic alkaloids , which also have antioxidant properties, which makes them medically interesting. About 10 to 14 percent of the population cannot break down Betalaine completely and excretes the dye in the urine (so-calledBeturia , as it occurs mainly after consuming beetroot ). Chemically, they are compounds of betalamic acid with various ligands. Depending on the ligand, the betalaines can be divided into the group of red-violet betacyans and the group of yellow betaxanthines .
Betalines can be distinguished from other similar water-soluble dyes by their spectral and electrophoretic properties and by the fact that they disintegrate in a hot, acidic environment.
In the 1960s, Hugo Wyler and others determined the basic structure of the Betalaine. The most important reactions of betalain biosynthesis were also elucidated during this time . Betalaine-producing plants are not able to convert flavan-3,4-diols into anthocyanidins, so anthocyanins and betalaine do not occur together.
biosynthesis
During the biosynthesis of betalines, the amino acid tyrosine is first converted to dopa by means of a hydroxylase and then converted to betalamic acid. This then reacts with cyclo-dopa on the one hand to form betanidin, the starting material for all betacyans, or on the other hand with various amino acids or amines to form various betaxanthines (see figure). The variety of betacyans results from different glycosidation and acylation of betanidine.
Betacyane
All betacyans have a purple hue. The best-known representative of betacyans is betanin (beet red), which is approved in the food and cosmetics industries . Below is a list of other well-known betacyans:
- Amaranthine. In the flowers and leaves of many foxtail plants .
- Betanidin. In flowers of Mesembryanthemum ( ice plant ) and Portulaca ( purslane family ).
- Bougainvillein-r-1. In bracts of bougainvillea .
- Celosianin II. In Celosia and species of goosefoot .
- Isobetanine. In fruits of opuntia and phyllocladia of Phyllocactus , as well as in flowers of Mesembryanthemum .
- Muscapurpurin. Part of the red color of the fruit body of the fly agaric .
- Prebetanin. In the skin of beets and fruits of the pokeweed family .
Betaxanthine
All betaxanthines are yellow. Here are several well-known members of the group:
- Dopaxanthin. In flowers of Glottiphyllum ( midday flowers ).
- Humilixanthin. In beets , ice cream plants , purslane plants and pokeweed plants .
- Muscaaurin I, II and VII. Part of the red color of the fruit body of the fly agaric , but also in beets .
- Portulacaxanthin I, II and III. In purslane family .
- Vulgaxanthin I and II. In beets .
literature
- Judith Hans: Cloning and characterization of flavonoid glucosyltransferases from Beta vulgaris L. Diss., Martin Luther University Halle-Wittenberg June 23, 2004 online version
- JB Harbourne: Phytochemical Dictionary. Taylor & Francis. 1998. ISBN 0748406204 , pp. 159ff.
- P. Schopfer, A. Brennecke: Plant physiology. Elsevier GmbH, Spektrum Akademischer Verlag 6th edition 2006, page 355 ff. ISBN 3-8274-1561-6
Individual evidence
- ↑ H. Wyler: The Betalaine . In: Chemistry in Our Time . tape 3 , no. 5 , 1969, p. 146–151 , doi : 10.1002 / ciuz.19690030504 ( PDF ( Memento of February 9, 2012 in the Internet Archive )).