Dysprosium (III) oxide
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
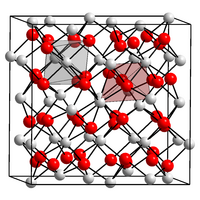
|
||||||||||||||||
| __ Dy 3+ __ O 2− | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Dysprosium (III) oxide | |||||||||||||||
| other names |
Dysprosia |
|||||||||||||||
| Ratio formula | Dy 2 O 3 | |||||||||||||||
| Brief description |
white, slightly hygroscopic powder |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 373.00 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
7.81 g cm −3 (25 ° C) |
|||||||||||||||
| Melting point |
2310 ° C |
|||||||||||||||
| boiling point |
3900 ° C |
|||||||||||||||
| solubility |
|
|||||||||||||||
| Refractive index |
1.9757 |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
Dysprosium (III) oxide is a chemical compound from the group of oxides .
history
In 1878 erbium ores were discovered which, in addition to this, also contained holmium and thulium (III) oxide . The French chemist Paul Émile Lecoq de Boisbaudran was able to isolate dysprosium (III) oxide while working with holmium (III) oxide in Paris in 1886 and thus discovered the element dysprosium .
Extraction and presentation
Dysprosium (III) oxide can be obtained by burning dysprosium in air.
properties
Dysprosium (III) oxide is a white, slightly hygroscopic powder that is insoluble in water and strongly magnetic. It has a cubic crystal structure. It reacts with acids to form cations, which in turn react weakly acidic in water.
use
Dysprosium (III) oxide is used for special purposes in glass, fluorescent materials, lasers and metal halide lamps. It will continue in the cermet for nuclear reactor - control rods used. As a dopant for Bariumtitanatoxid - capacitors , it is used high capacity for small capacitors. Its magnetic properties make it interesting for motors and generators.
Individual evidence
- ↑ a b c d Reade: Dysprosium oxide ( page no longer available , search in web archives ) Info: The link was automatically marked as defective. Please check the link according to the instructions and then remove this notice.
- ↑ a b c d e data sheet Dysprosium (III) oxide, ≥99.99% trace metals basis from Sigma-Aldrich , accessed on March 1, 2012 ( PDF ).
- ^ A b David R. Lide: CRC Handbook of Chemistry and Physics, 87th Edition . 2006, ISBN 978-0-8493-0487-3 , pp. 4-63 .
- ↑ a b Data sheet dysprosium oxide (PDF) from Merck , accessed on March 1, 2012.
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Index of Refraction of Inorganic Crystals, pp. 10-246.
- ^ Paul Émile Lecoq de Boisbaudran: L'holmine (ou terre X de M Soret) contient au moins deux radicaux métallique (Holminia contains at least two metal) . In: Comptes Rendus . 143, 1886, pp. 1003-1006.
- ^ A b Robert E. Krebs: "Dysprosium". The History and Use of our Earth's Chemical Elements . Greenwood Press, 1998, ISBN 0-313-30123-9 , pp. 234-235 .
- ↑ G. Adachi, Nobuhito Imanaka and ZC Kang: Binary rare earth oxides . Springer Netherlands, 2004, ISBN 978-1-4020-2568-6 ( limited preview in Google book search).
- ↑ Periodic table of the elements online: acid-base behavior: dysprosium
- ^ John Emsley: Nature's building blocks: an AZ guide to the elements . Oxford University Press, 2003, ISBN 978-0-19-850340-8 , pp. 131 ( limited preview in Google Book search).
- ^ Steven Chu: Critical Materials Strategy . Diane Publishing Books, 2010, ISBN 978-1-4379-4418-1 , pp. 77 ( limited preview in Google Book search).

