Ethyl isocyanate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
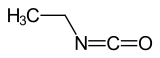
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Ethyl isocyanate | |||||||||||||||
| other names |
Ethyl isocyanate |
|||||||||||||||
| Molecular formula | C 3 H 5 NO | |||||||||||||||
| Brief description |
colorless liquid with a pungent odor |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 71.08 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
0.91 g cm −3 |
|||||||||||||||
| Melting point |
<−50 ° C |
|||||||||||||||
| boiling point |
59-61 ° C |
|||||||||||||||
| Vapor pressure |
147 hPa (20 ° C) |
|||||||||||||||
| solubility |
decomposes in water |
|||||||||||||||
| Refractive index |
1.380 (20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
Ethyl isocyanate is a chemical compound belonging to the isocyanate group . It has the constitutional formula CH 3 –CH 2 –NCO.
Extraction and presentation
Ethyl isocyanate can be obtained by reacting triphosgene with ethylamine hydrochloride in xylene.
properties
Ethyl isocyanate is a highly flammable, volatile, colorless liquid with a pungent odor that decomposes in water.
use
Ethyl isocyanate is used in the manufacture of drugs and pesticides.
safety instructions
The vapors of ethyl isocyanate can form an explosive mixture with air ( flash point −10 ° C).
Individual evidence
- ↑ a b c d e f g h i j k Entry on ethyl isocyanate in the GESTIS substance database of the IFA , accessed on January 8, 2020(JavaScript required) .
- ↑ Data sheet Ethyl isocyanate, 98% from AlfaAesar, accessed on December 22, 2016 ( PDF )(JavaScript required) .
- ↑ Entry on ethyl isocyanate in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on December 22, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ One-step synthesis of ethyl isocyanate manufacturing method: One-step synthesis of ethyl isocyanate manufacturing method ( Memento of the original from December 23, 2016 in the Internet Archive ) Info: The archive link has been inserted automatically and has not yet been checked. Please check the original and archive link according to the instructions and then remove this notice. , Access date: December 22, 2016.
- ↑ Richard P. Pohanish: Sittig's Handbook of Toxic and Hazardous Chemicals and Carcinogens, 5th Edition . William Andrew, 2008, ISBN 978-0-8155-1904-1 , pp. 1204 ( limited preview in Google Book search).



