Fallot tetralogy
| Classification according to ICD-10 | |
|---|---|
| Q21.3 | Fallot's tetralogy Ventricular septal defect with pulmonary stenosis or atresia, dextroposition of the aorta, and hypertrophy of the right ventricle |
| ICD-10 online (WHO version 2019) | |
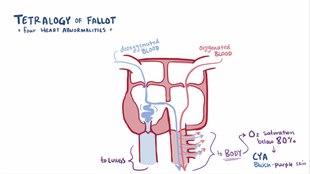
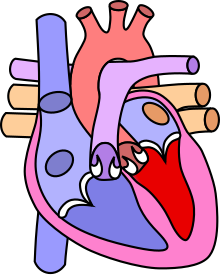
The Fallot tetralogy (also Fallot's tetralogy ) is a congenital heart malformation that accounts for about 6–7% of congenital heart defects. It consists of four components (hence tetralogy ): a pulmonary stenosis , a ventricular septal defect (defect in the cardiac septum), an aorta that rides above the cardiac septum, and a subsequent right heart hypertrophy . The abbreviation TOF , which is also used by medical professionals in German, is derived from the English Tetralogy of Fallot . In about a third of the cases, other malformations occur in addition to the heart changes, especially in the digestive tract and crevices.
history
The name of this heart defect goes back to the French pathologist Étienne-Louis Arthur Fallot as a descriptor (1888). The first description of the Fallot tetralogy comes from Stensen in 1672, another by Eduard Sandifort (1777). The pathologist Carl von Rokitansky also described this malformation (1876). The English surgeon Thomas Bevill Peacock (1812-1882) described it in 1858 in the first detailed treatise on congenital heart malformations.
In 1945, Blalock and Taussig surgically corrected a Fallot tetralogy for the first time by creating a connection ( shunt ) between the subclavian artery and the pulmonary artery . In 1946, Potts et al. Established an anastomosis between the aorta and the left pulmonary artery, and in 1962 Waterson between the aorta and the right pulmonary artery.
Emergence
There are actually no four separate defects in this malformation, but one, namely a forward displacement of the infundibular septum of the right ventricle.
At the end of the fifth week of development, the heart's outflow path still arises from both halves of the heart. The formation of endocardial bulges in the conus cordis creates the septum aorticopulmonale, which divides the conus cordis into right and left outflow tract ( aorta and pulmonary trunk ). However, if the aorticopulmonary septum moves forward, the ventricular outflow area narrows ( pulmonary stenosis ), but the septum is still open ( ventricular septal defect ). The aorta now arises from both heart chambers (riding aorta). The pulmonary stenosis causes high blood pressure ( arterial hypertension ) in the right ventricle . So that enough blood can still be pumped out of the heart, the chamber wall enlarges ( hypertrophied right ventricle).
In 10% of cases, the Fallot tetralogy is associated with a chromosomal aberration , most often with Edwards syndrome or Down syndrome .
The malformations
The following malformations form the clinical picture:
Pulmonary stenosis
Pulmonary stenosis as a narrowing of the exit tract from the right ventricle to the lungs . The individual parts of this outflow tract (part near the heart, valve ring with crescent-shaped valves (semilunar) and part near the lungs) can be narrowed to different degrees. The actual pulmonary valve is often malformed and has only two crescent valves in over 60% of cases. Therefore, the severity of the valve narrowing can vary widely. The pulmonary vascular system is - as a rule - underdeveloped, corresponding to the constrictions in the entire outflow tract.
Ventricular septal defect
The ventricular septal defect is a hole in the septum between the chambers of the heart, which in the Fallot tetralogy is not a lack of substance in the ventricular septum, but rather a result of the malformation of the heart. It is usually located directly below the right ventricular outflow tract (pulmonary valve) and near the aortic valve (outlet valve for the circulatory system) and the tricuspid valve (valve between the right main chamber and the right atrium).
Crossing the aorta
Due to a deficient rotation of the pulmonary vessel in the development of the heart, the aortic root remains positioned in its relationship to the right ventricle, so that a " crossing of the aorta " results over the ventricular septal defect.
Right ventricular hypertrophy
Right ventricular hypertrophy, an increase in muscle mass of the right heart that hinders the functioning of the heart due to chronic overload, can be slight at first, but increase over time.
Consequences for the cardiovascular system
Due to the narrowing of the pulmonary outflow tract, the oxygen-poor (venous) blood returning from the body via the right auricle only partially reaches the lungs, where it is enriched with oxygen. A correspondingly larger proportion escapes via the large ventricular septal defect and flows back into the body directly via the aorta ( large body artery ) , bypassing the pulmonary circulation . Such a transfer of blood from the right heart directly into the body's circulation is called a right-left shunt .
Symptoms
The mixture of oxygen-poor blood via the shunt with the oxygen-rich blood returning from the lungs causes cyanosis . This can be seen in the blue discoloration of the skin and mucous membranes , mainly on the mouth and hands / feet, due to the lack of oxygen in the body's circulation. It can be more or less pronounced depending on the amount of blood spilled and may only be visible after exercise (drinking or screaming in young children). If the cyanosis is low, one speaks of a “pink fallot”, in which velvety, deep red mucous membranes can be seen. Severe cyanosis occurring during the first months of life (after the ductus arteriosus has been closed) suggests a deformity of the heart when looking at the patient . On palpation, a systolic whirring can be felt over the base of the heart.
The body reacts to the lack of oxygen in the blood by increasing the number of erythrocytes (red blood cells). In this way, the oxygen deficit can be compensated within certain limits. But the flow properties of the blood are also changed and small clots can form, which can trigger an embolism . The growth of the small vessels on the fingers and toes also increases. So-called drumstick fingers and toes as well as watch glass nails can develop. Because of the impaired flow properties of the blood, the children should always be offered enough to drink. The existing right ventricular hypertrophy (enlargement of the right ventricle) can be recognized during percussion (wooden shoe shape of the heart).
Hypoxemic seizures
A peculiarity of the Fallot tetralogy are hypoxemic seizures. They only occur in a small proportion of children, but are rarely seen in children with other cyanotic heart defects. With such an attack, the children become restless and anxious, the cyanosis increases, the heart rate rises to values between 140 and 160 beats / minute. The children can appear gray-pale-blue and the consciousness is clouded to the point of unconsciousness. The seizures usually develop without any recognizable external cause and the cause is not always clearly recognizable.
An attack of hypoxemia requires emergency treatment. Its appearance usually requires an early operation. Fresh air and calming down are suitable as first aid in domestic conditions. In the clinic, an attack is treated with oxygen, drug sedation (such as opioids ) and beta blockers (such as propranolol ; can have a prophylactic effect).
Diagnosis
During auscultation, a loud ( also spindle-shaped in phonocardiography ) systolic noise can be heard above the base of the heart , the loudest to the left of the sternum. Today, echocardiography and, more rarely, cardiac catheterization are usually available for diagnosis . The related clinical picture of a common arterial trunk can be ruled out if the 2nd heart tone is split.
therapy
Therapy consists of a surgical correction. The aim is to create a normal anatomy. The pulmonary stenosis is widened and the ventricular septal defect is closed with a patch - usually made of pericardium or Goretex - so that the aorta receives only oxygen-rich blood from the left ventricle (heart chamber). The aim is to have one-step surgery in the first year of life. However, it depends on the individual factors in the patient whether this procedure is possible. In the past, a Blalock-Taussig anastomosis was often created in order to reach a higher age for the correction . In the case of special constellations, it may still be indicated today to precede the main operation with a palliative operation by creating an aorto-pulmonary shunt. Most often, a modified Blalock-Taussig shunt in the form of a Goretex prosthesis is placed between the brachiocephalic trunk and the right pulmonary artery or a central shunt between the aorta and pulmonary artery. This operation is considered to be a lighter procedure and is usually performed without the use of the heart-lung machine . This increases the blood flow to the lungs and thus improves the oxygen supply. The shunt is closed again during the subsequent final operation. In some cases, the operation can be postponed for a few weeks or months in an infant as part of a cardiac catheter intervention in the form of expanding the pulmonary stenosis with a balloon.
Treatment results and check-ups
As a rule, there is no complication after the operation and the heart chambers can develop normally. Regular checkups (every six to twelve months, for life) by the pediatric cardiologist should definitely be carried out in order to observe any remaining minor pulmonary stenosis or leakage of the valve. In many cases, these can currently be treated with catheter intervention by inserting a new biological heart valve in the position of the pulmonary valve without a new operation. During these check-ups, attention is also paid to any cardiac arrhythmias . A Endokarditisprophylaxe will have to live long observed.
It is to be expected that the children operated on today will develop normally and that they will achieve normal physical resilience and a good quality of life in adulthood. However, as with all complex congenital heart defects, the course can be different. Previously operated patients in whom the corrective operation was only performed in childhood or adolescence for technical reasons (lack of early diagnosis and operation options) may not be comparable to the children who are operated on today.
facts and figures
- The spontaneous course depends on the extent of the blood circulation in the lungs. People with this disease (without surgery) have an average life expectancy of 12 years, 95% die before the age of 40.
- Sex ratio: male: female = 1.4: 1
- About 15% of patients have a microdeletion on chromosome 22q11 .
See also
literature
- S2k guideline Fallot tetralogy of the German Society for Pediatric Cardiology (DGPK). In: AWMF online (as of 2013-2018)
- F. Bailliard, RH Anderson: Tetralogy of Fallot. In: Orphanet Journal of Rare Diseases. Volume 4, 2009, p. 2, ISSN 1750-1172 . doi: 10.1186 / 1750-1172-4-2 . PMID 19144126 . PMC 2651859 (free full text). (Review).
- EA Shinebourne, SV Babu-Narayan, JS Carvalho: Tetralogy of Fallot: from fetus to adult. In: Heart. Volume 92, Number 9, September 2006, pp. 1353-1359, ISSN 1468-201X . doi: 10.1136 / hrt.2005.061143 . PMID 16908723 . PMC 1861206 (free full text). (Review).
- FA Pigula et al .: Repair of tetralogy of Fallot in neonates and young infants. In: Circulation. Volume 100, Number 19 Suppl, November 1999, pp. II157-II161, ISSN 0009-7322 . PMID 10567296 .
- Klaus Holldack, Klaus Gahl: Auscultation and percussion. Inspection and palpation. Thieme, Stuttgart 1955; 10th, revised edition, ibid 1986, ISBN 3-13-352410-0 , pp. 187-190 and 196 f.
Web links
- Fallot tetralogy. In: Orphanet (Rare Disease Database).
- herzklick.de: Animated heart defect description of a Fallot tetralogy
Individual evidence
- ↑ a b Christof Sohn at al .: Ultrasound in gynecology and obstetrics . Georg Thieme, Stuttgart 2003, ISBN 978-3-13-101972-1 , p. 195.
- ↑ a b Christof Schmid: Guide to Pediatric Heart Surgery. Springer, Berlin 2013, ISBN 978-3-662-12258-7 , p. 47.
- ^ Klaus Holldack, Klaus Gahl: Auscultation and percussion. Inspection and palpation. Thieme, Stuttgart 1955; 10th, revised edition ibid 1986, ISBN 3-13-352410-0 , pp. 100 and 187.
- ↑ TB Peacock: On malformations [...] of the human heart. London 1858.
- ↑ Barbara I. Tshisuaka: Peacock, Thomas Bevill. In: Werner E. Gerabek , Bernhard D. Haage, Gundolf Keil , Wolfgang Wegner (eds.): Enzyklopädie Medizingeschichte. De Gruyter, Berlin / New York 2005, ISBN 3-11-015714-4 , p. 1119.
- ↑ Christof Schmid: Guide to Pediatric Heart Surgery. 2013, p. 48.
- ↑ Fallotsche Tetralogy S2k German Society for Pediatric Cardiology (DGPK) , AWMF, 2013-2018 (archived version accessed on February 16, 2020)
- ^ Klaus Holldack, Klaus Gahl: Auscultation and percussion. Inspection and palpation. Thieme, Stuttgart 1955; 10th, revised edition ibid 1986, ISBN 3-13-352410-0 , p. 189 f. and 196 f.
- ↑ Luca Oechslin, Roberto Corti, Matthias Greutmann, Oliver Kretschmar, Oliver Gaemperli: Percutaneous pulmonary valve implantation in grown-up congenital heart disease patients: Insights from the Zurich experience . In: Journal of Interventional Cardiology . tape 31 , no. 2 , 2018, ISSN 1540-8183 , p. 251-260 , doi : 10.1111 / joic.12477 ( wiley.com [accessed February 6, 2019]).
- ↑ a b c Herold, Gerd : Innere Medizin, Cologne, 2015, p. 193, ISBN 978-3-9814660-8-9 .