Ferroelectric
Ferroelectricity (or polarization catastrophe ) describes the phenomenon that substances with an electric dipole moment change the direction of spontaneous polarization when an external electric field is applied .
Ferroelectricity was formerly known as Seignette Electricity (rarely as Seignettesalzelektrizität ), referred to as it the Rochelle salt was discovered (potassium sodium tartrate).
properties
Ferroelectricity only occurs in crystals in which the crystalline symmetry allows a polar axis. As a result, the shifting of differently charged ions in the crystal lattice leads to spontaneous polarization. The electrical polarization in ferroelectrics can be reversed by applying an external voltage . Ferroelectric materials are always pyroelectric and thus also piezoelectric .
The prefix “Ferro-” does not refer to a property of iron in ferroelectrics, but to the analogy to ferromagnetism . As with the ferromagnetic magnetization, so with ferroelectrics the polarization disappears at high temperatures (the ferroelectric Curie temperature ) - the material is then paraelectric . Above this temperature, the electrical susceptibility follows the Curie-Weiss law analogous to the magnetic susceptibility . When the material cools down, a phase transition takes place when the temperature falls below the Curie temperature, which usually coincides with a structural change (reduction in crystal symmetry) and the material becomes ferroelectric again . The relative permittivity increases significantly at lower temperatures; at 4 K it is often 10 to 20 times higher than the value at room temperature and can reach values above 10 6 .
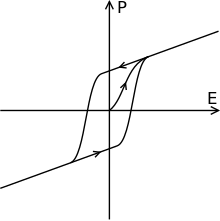
Ferroelectric crystals form domains , i.e. areas with the same polarization direction. The direction of polarization changes from domain to domain in the area of a few atomic layers in which the polarization disappears. The ferroelectric domain walls are only a few nanometers wide. In contrast, in ferromagnetism, the orientation of the magnetization changes gradually over a range of 10 nm and more. Because of the narrower domain walls, domains with different orientations in ferroelectric thin films can have a higher density than in ferromagnetic thin films. It is therefore hoped that the maximum information density in the development of ferroelectric storage media will be higher than that of ferromagnetic ones. The polarization can be reversed by applying an external electric field , as shown in the adjacent sketch, and follows a hysteresis curve .
But there are also ferrielectric and antiferroelectric substances, similar to magnetic materials. The superposition of several sub-grids of ordered electrical dipoles characterizes an antiferroelectric order. This superposition has an opposite but equally strong electrical polarization. Accordingly, the macroscopic total polarization .
Every antiferroelectric substance has a Curie temperature . Above this temperature, both partial grids are completely equivalent and unpolarized. However, it can happen that the antiparallel dipole moments of the partial lattices do not cancel each other out completely. As a result, a finite polarization results . In this case it is ferric electricity .
application
Ferroelectrics are used to manufacture highly precise mechanical actuators (sliding elements ). Using the inverse piezoelectric effect, displacements with a resolution of less than an atomic diameter are possible. They are used, for example, in atomic force microscopes , scanning tunneling microscopes or other scanning probe microscopes .
Ferroelectrics usually have a high to very high relative permittivity ( ) in the range between 100 and 100,000, which is why they are used as a material for ceramic capacitors with high volume capacities. They are increasingly replacing the electrolytic capacitors and are characterized by low equivalent series resistances and inductances (ESR and ESL). The disadvantages, however, are the strong temperature dependence, the large tolerances and the high dielectric loss factors . This high permittivity also makes them interesting in semiconductor technology , where high capacities are required in the smallest of spaces for smaller memory circuits ( RAM ). The main advantage of using so-called FeRAM ( ferroelectric RAM ) is that it does not lose its state of charge compared to the DRAMs that are currently mainly used (2008) . In this case, one speaks of non-volatile memories ( NVRAM ). Another area of research is the use of ferroelectrics as so-called high-k dielectrics . Perovskites in particular could in future replace silicon dioxide as the gate dielectric of field effect transistors in integrated circuits . Due to the permanent polarizability, they are still suitable as electrets , e.g. B. in sensors and microphones.
Examples
The best-known ferroelectrics are ion crystals with a perovskite structure such as:
- Barium titanate BaTiO 3 (Abbreviation: BTO)
- Lead zirconate titanate Pb (Zr x Ti 1-x ) O 3 (PZT) - is often used for the production of piezomechanical actuators.
The following substances are also ferroelectric, but sometimes only in the form of thin layers :
- Strontium bismuth tantalate SrBi 2 Ta 2 O 9 (SBT)
- Bismuth titanate Bi 4 Ti 3 O 12 (BIT, also misleading BTO)
- Bismuth lanthanum titanate Bi 4-x La x Ti 3 O 12 (BLT)
- Bismuth titanate niobate Bi 3 TiNbO 9 (BTN)
- Strontium titanate SrTiO 3 (STO)
- Barium Strontium Titanate Ba x Sr 1-x TiO 3 (BST)
- Sodium nitrite NaNO 2
- Lithium Niobate LiNbO 3
- Potassium Sodium Tartrate Tetrahydrate ( Seignette Salt ) KNaC 4 H 4 O 6 · 4 H 2 O
Hexagonal manganates RMnO 3 with R = Sc, Y, In, Dy, Ho, Er, Tm, Yb, Lu.
Organic ferroelectrics were also found:
- 1,1-di (carboxymethyl) cyclohexane or
- Triglycine sulfate (CH 2 NH 2 COOH) 3 · H 2 SO 4 (TGS)
- Polyvinylidene fluoride (PVDF) , or the copolymer poly (vinylidene fluoride-trifluoroethylene) P (VDF-TrFE)
Well-known antiferroelectrics are ammonium dihydrogen phosphate (ADP) or some substances with a perovskite structure such as:
- Lead zirconate ( )
- Sodium niobate
- Lead affnat
literature
- Hans Fischer: Materials in electrical engineering. 2nd edition, Carl Hanser Verlag, Munich / Vienna, 1982 ISBN 3-446-13553-7 .
- Horst Stöcker: Pocket book of physics. 4th edition, Verlag Harry Deutsch, Frankfurt am Main 2000, ISBN 3-8171-1628-4 .
- Rudolf Gross, Achim Marx: Solid State Physics. 3rd edition, De Gruyter Studium, Berlin / Boston 2018, ISBN 978-3-11-055822-7 .
Individual evidence
- ↑ Seignette electricity. In: Brockhaus. 2003
- ↑ Markus Aspelmeyer: Influence of external fields on the structure and interface morphology of thin ferroelectric films . ( Archive server deposit.d-nb.de - dissertation, Munich, Ludwig-Maximilians-Universität, 2002).
- ^ LJ Gauckler and K. Conder: Ceramics II. Lecture notes . ETH Zurich, p. 37 ( PDF, 7.1 MB ).
- ↑ Nakamura, K. and Wada, Y. (1971). Piezoelectricity, pyroelectricity, and the electrostriction constant of poly (vinylidene fluoride). Journal of Polymer Science Part A-2: Polymer Physics, 9 (1): 161-173.