Indium (I, III) bromide
| Crystal structure | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||
| __ In + __ In 3+ __ Br - | |||||||||||||
| Space group |
Pnna (No. 52) |
||||||||||||
| General | |||||||||||||
| Surname | Indium (I, III) bromide | ||||||||||||
| other names |
|
||||||||||||
| Ratio formula | InBr 2 | ||||||||||||
| Brief description |
light yellow solid |
||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 274.63 g mol −1 | ||||||||||||
| Physical state |
firmly |
||||||||||||
| density |
4.2 g cm −3 |
||||||||||||
| Melting point |
197.1 ° C |
||||||||||||
| boiling point |
630 ° C (decomposition) |
||||||||||||
| solubility |
reacts with water |
||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Indium (I, III) bromide is an inorganic chemical compound of indium from the group of bromides .
Extraction and presentation
Indium (I, III) bromide can be obtained by reacting indium with bromine at 300 to 400 ° C or with indium (III) bromide at 350 ° C.
It also occurs in the waste from the production of semiconductors.
properties
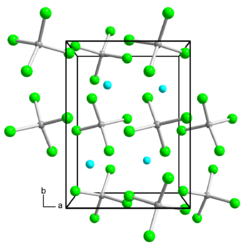
Indium (I, III) bromide is a light yellow diamagnetic solid. It can be separated into indium (I) bromide and indium (III) bromide (ether-soluble) by ether extraction . Indium (I, III) bromide (In [InBr 4 ]) is a mixed-valence indium I – III bromide of the gallium (I, III) chloride type with an orthorhombic crystal structure and crystallizes in the space group Pnna (space group no. 52) with the lattice parameters a = 798.6 pm, b = 1038.5 pm, c = 1042.5 pm and eight formula units per unit cell .
Individual evidence
- ↑ a b c d e Georg Brauer (Ed.), With the collaboration of Marianne Baudler u. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6 , p. 868.
- ↑ WebElements: WebElements Periodic Table of the Elements | Indium | indium dibromide , accessed Tuesday, March 18, 2014
- ↑ a b ya. M. Grushko: Handbook of Dangerous Properties of Inorganic And Organic Substances in ... CRC Press, 1992, ISBN 0-8493-9300-0 , p. 52 ( limited preview in Google Book search).
- ^ William M. Haynes: CRC Handbook of Chemistry and Physics, 93rd Edition . CRC Press, 2012, ISBN 1-4398-8049-2 , pp. 4–67 ( limited preview in Google Book Search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Thomas Staffel, Gerd Meyer: The mono-, sesqui-, and dibromides of indium: InBr, In2Br3, and InBr2. In: Journal of Inorganic and General Chemistry. 552, 1987, pp. 113-122, doi : 10.1002 / zaac.19875520913 .

