Mineralocorticoid receptor
| Mineralocorticoid receptor | ||
|---|---|---|
|
||
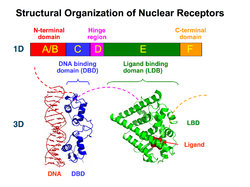
| Schematic representation of the structure of nuclear receptors. | ||
| Properties of human protein | ||
| Mass / length primary structure | 984 amino acids | |
| Secondary to quaternary structure | Heterooligomer (unalloyed) | |
| Isoforms | 1, 2, 3, delta | |
| Identifier | ||
| Gene name | NR3C2 | |
| External IDs | ||
| Occurrence | ||
| Parent taxon | Vertebrates | |
The mineralocorticoid receptor (aldosterone receptor, MR, NR3C2) is a nuclear receptor that is activated by binding the steroid hormones aldosterone or cortisol and then acts as a transcription factor . It is involved in the regulation of the water-electrolyte balance in the kidneys and large intestine . It also has numerous other functions in various other tissues. Mutations in NR3C2 - gene can a change in the activity of the transcription factor entail; Loss-of-function mutations are then the cause of pseudohypoaldosteronism type 1 and gain-of-function mutations lead to congenital high blood pressure .
structure
The cloning of the mineralocorticoid receptor gene was first described in 1987. The MR gene is located on chromosome 4, region q31.1, and comprises approximately 450 kB. Within the superfamily of nuclear receptors, the mineralocorticoid receptor belongs to subfamily 3 (estrogen receptor-like), group C.
The mineralocorticoid receptor is made up of three functional domains: The N-terminal domain (NTD) interacts with cofactors independently of ligands and with the ligand-binding domain (LBD) depending on a ligand. The ligand-binding domain shows a high structural similarity within the steroid receptor family.
Mechanism of action
The mechanism of action of the mineralocorticoid receptor corresponds to that of a transcription factor. In the absence of a ligand , the receptor lies in the cytoplasm as a complex with heat shock proteins . After binding a ligand, MR forms dimers and translocates into the cell nucleus . There it binds to a specific DNA sequence (AGAACANNNTGTTCT) in the promoter region of target genes and thus regulates their transcription. Aldosterone and cortisol bind to MR with a comparable affinity, with cortisol in the plasma in about 1000 times higher concentrations. The (relative) selectivity of MR for aldosterone is achieved through various mechanisms: The co-expressed enzyme 11-beta-hydroxsteroid dehydrogenase (11-βHSD2) converts cortisol into the inactive form cortisone and thus prevents MR activation. In addition, the interaction of receptor subunits with one another and with numerous co-activators and repressors influences the transcriptional activity of the receptor. A described target gene of the mineralocorticoid receptor is serum glucocorticoid-regulated kinase 1 ( Sgk1 ), a protein kinase that is considered the starting point for various cellular signal cascades.
Aldosterone also mediates its effect independently of MR via so-called non-genomic signaling pathways which cannot be blocked by transcription inhibitors and antagonists at the mineralocorticoid receptor.
function
The mineralocorticoid receptor is expressed in a variety of tissues, including the kidney and colon, heart, and central nervous system, where it performs different functions. The function in epithelial cells is distinguished from that in non-epithelial cells.
epithelium
The mineralocorticoid receptor is involved in the regulation of the water-electrolyte balance in epithelial cells in the kidneys and large intestines . Activation of the receptor leads to increased expression of ion channels and transporters such as the amiloride-sensitive sodium channel (ENaC) and the basolateral sodium-potassium-ATPase . This facilitates the transport of sodium across the epithelium, which leads to increased water reabsorption. At the same time, more potassium is excreted.
Non-epithelia
Cardiovascular system
Increased activation of the mineralocorticoid receptor leads to high blood pressure on the one hand, but also has a direct effect on the heart, where increased aldosterone levels can cause pathological enlargement of the heart and connective tissue storage. The molecular mechanisms that lead to these effects have not yet been conclusively clarified. There is evidence that the mineralocorticoid receptor in macrophages is pathogenetically involved in the abovementioned proliferation of connective tissue.
Central nervous system
Mineralocorticoid receptors are found in high density in the central nervous system, including in the hypothalamus. There is some evidence that central MRIs are involved in the control of the sympathetic nervous system and blood pressure.
pharmacology
Antagonists
Spironolactone is a competitive antagonist at the mineralocorticoid receptor and belongs to the group of potassium-sparing diuretics . The substance has been in clinical use for a long time and is used, among other things, in the therapy of ascites . A more modern substance is eplerenone , which, due to its higher specificity for MR, has a more favorable side effect profile compared to other steroid receptors. At the beginning of the 21st century, two large clinical studies, RALES and EPHESUS, showed a life-prolonging effect of aldosterone antagonists in chronic heart disease and thus contributed to the increasing importance of the substances. In 2011 these results were confirmed for patients with mild heart failure. The clinically used antagonists spironolactone and eplerenone block the mineralocorticoid receptor in the cardiovascular system, but also in the kidneys. This can lead to electrolyte imbalances and a deterioration in kidney function. We are therefore working on the development of various alternative drugs based on a modified principle of action or a modified structure. Due to its different pharmacokinetics and distribution, the substance finerenone shows a higher activity in the heart compared to the kidneys and could thus avoid these undesirable effects, but is still in clinical testing.
Agonists
The synthetic mineralocorticoid fludrocortisone , which is used in primary adrenal insufficiency in the context of adrenogenital syndromes , is used in clinical applications.
literature
- S Viengchareun, D Le Menuet, L Martinerie, M Munier, L Pascual-Le Tallec, M Lombès: The mineralocorticoid receptor: insights into its molecular and (patho) physiological biology . In: Nucl Recept Signal. 30, 5, 2007, p. E012, PMID 18174920
- L Pascual-Le Tallec, M Lombès: The mineralocorticoid receptor: a journey exploring its diversity and specificity of action . In: Mol Endocrinol . 19 (9), 2005, pp. 2211-2221, PMID 15802372
- JM Connell, E Davies: The new biology of aldosterone . In: J Endocrinol . 186 (1), 2005, pp. 1-20, PMID 16002531
- PJ Fuller, MJ Young: Mechanisms of mineralocorticoid action . In: Hypertension . 46 (6), 2005, pp. 1227-1235, PMID 16286565
Web links
- MC Zennaro: Pseudohypoaldosteronism, renal, type 1. In: orpha.net. Orphanet, January 2009, accessed December 19, 2010 .
Individual evidence
- ↑ Swiss Institute of Bioinformatics (SIB): PROSITE documentation PDOC00031. Retrieved September 20, 2011 .
- ↑ UniProt P08235
- ↑ Jeffrey L. Arriza, Cary Weinberger, Gail Cerelli, Tom M. Glaser, Barbara L. Handelin, David E. Housman, Ronald M. Evans: Cloning of Human Mineralocorticoid Receptor Complementary DNA: Structural and Functional Kinship with the Glucocorticoid Receptor . In: Science . 237 (4812), 1987, pp. 268-275
- ↑ Entrez Gene ID 4306
- ^ A unified nomenclature system for the nuclear receptor superfamily . Nuclear Receptors Nomenclature Committee In: Cell . 97 (2), 1999, pp. 161-163, PMID 10219237 doi: 10.1016 / S0092-8674 (00) 80726-6
- ^ A b L. Pascual-Le Tallec, M Lombès: The mineralocorticoid receptor: a journey exploring its diversity and specificity of action . In: Mol Endocrinol . 19 (9), 2005, pp. 2211-2221, PMID 15802372
- ↑ CG Brilla, KT Weber: Mineralocorticoid excess, dietary sodium, and myocardial fibrosis . In: J Lab Clin Med . 120 (6), 1992, pp. 893-901, PMID 1453111
- ↑ AJ Rickard, J Morgan, G Tesch, JW Funder, PJ Fuller, MJ Young: Deletion of mineralocorticoid receptors from macrophages protects against deoxycorticosterone / salt-induced cardiac fibrosis and increased blood pressure . In: Hypertension . 54 (3), 2009, pp. 537-543. PMID 19635989
- ↑ MG Usher, SZ Duan, CY Ivaschenko, RA Frieler, S Berger, G Schütz, CN Lumeng, RM Mortensen: Myeloid mineralocorticoid receptor controls macrophage polarization and cardiovascular hypertrophy and remodeling in mice . In: J Clin Invest . 120 (9), 2010, pp. 3350-3364, PMID 20697155
- ↑ JC Geerling, AD Loewy: Aldosterone in the brain . In: Am J Physiol Renal Physiol. 297 (3), 2009, pp. F559-576, PMID 19261742
- ↑ B Pitt, F Zannad, WJ Remme, R Cody, A Castaigne, A Perez, J Palensky, J Wittes: The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators . In: N Engl J Med. 2,341 (10), 1999, pp. 709-717, PMID 10471456
- ^ B Pitt, W Remme, F Zannad, J Neaton, F Martinez, B Roniker, R Bittman, S Hurley, J Kleiman, M Gatlin: Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction . In: N Engl J Med . 3, 348 (14), 2003, pp. 1309-1321, PMID 12668699
- ↑ F Zannad, JJ McMurray, H Krum, DJ van Veldhuisen, K Swedberg, H Shi, J Vincent, SJ Pocock, B Pitt: EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms . In: N Engl J Med. Jan 2011, 6, 364 (1), pp. 11-21, PMID 21073363
- ^ A Lother, M Moser, C Bode, RD Feldman, L Hein: Mineralocorticoids in the Heart and Vasculature: New Insights for Old Hormones. In: Annu. Rev. Pharmacol. Toxicol. Feb 2015. 55: 8.1-8.24. doi: 10.1146 / annurev-pharmtox-010814-124302 .
- ↑ B Pitt B, L Kober, P Ponikowski, M Gheorghiade, G Filippatos, H Krum, C Nowack, P Kolkhof, SY Kim, F Zannad. Safety and tolerability of the novel non-steroidal mineralocorticoid receptor antagonist BAY 94-8862 in patients with chronic heart failure and mild or moderate chronic kidney disease: a randomized, double-blind trial. In: Eur Heart J. Aug 2013, 34 (31), pp. 2453-2463. PMID 23713082


