Paraoxon
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
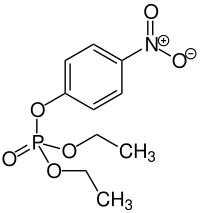
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Paraoxon | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 10 H 14 NO 6 P | |||||||||||||||
| Brief description |
flammable liquid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| Drug information | ||||||||||||||||
| ATC code | ||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 275.2 g mol −1 | |||||||||||||||
| density |
1.27 g cm −3 (25 ° C) |
|||||||||||||||
| boiling point |
169-170 ° C at 1.33 hPa |
|||||||||||||||
| solubility |
heavy in water (2.4 g l −1 at 25 ° C) |
|||||||||||||||
| Refractive index |
1.5096 |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
Paraoxon , synonym: E 600 , is an ester of phosphoric acid (see also: phosphoric acid ester ). The synonym is not an E number in the sense of food additives , but a number that was assigned when the compound was developed.
properties
Paraoxon is a colorless liquid . It is an active metabolite of parathion and therefore also an insecticide . In the liver as well as in many other tissues, Paraoxon can be formed from Parathion through oxidation through several intermediate stages. Monooxygenases in the microsomes of the endoplasmic reticulum are responsible for this . Paraoxon irreversibly inhibits the enzyme acetylcholinesterase . The high toxicity is due to the inhibition of acetylcholinesterase, which plays an important role in the body in the central nervous system (CNS). This makes paraoxon a neurotoxin .
Individual evidence
- ↑ a b c d e f Entry on Paraoxon in the GESTIS substance database of the IFA , accessed on February 1, 2016(JavaScript required) .
- ↑ a b Paraoxon data sheet from Sigma-Aldrich , accessed on April 18, 2011 ( PDF ).
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Physical Constants of Organic Compounds, pp. 3-410.
- ↑ Harun Parlar, Daniela Angerhöfer: Chemical Ecotoxicology . Springer Verlag, 2nd edition, 1995, p. 142, ISBN 3-540-59150-8 .

