Spiroplasma
| Spiroplasma | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
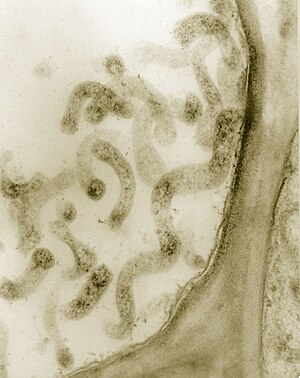
Spiroplasma kunkelii in cells of the phloem . |
||||||||||||
| Systematics | ||||||||||||
|
||||||||||||
| Scientific name | ||||||||||||
| Spiroplasm | ||||||||||||
| Saglio et al. 1973 |
Spiroplasma ( Spiroplasma ) (derived from the two ancient Greek words σπεῖρα spéïra "spiral" and πλάσμα plásma "shaped") are a genus of small bacteria without a cell wall in the class of Mollicutes . All spiroplasms live parasitically in plants, arthropods or rodents or are saprophytic .
description
Species of the genus Spiroplasma have pleomorphic , rounded shapes and agile helical or branched, non-helical filaments. The diameter is approximately 120 nm and the length 2 to 4 μm or more. The genome size is 10 9 daltons . The GC content is between 25 and 26.4 mol%.
Pathogenicity
The transmission of phytopathogenic forms occurs through cicadas as vectors . The infection is limited to the phloem . The connection between spiroplasma and numerous plant diseases has been described. However, their pathogenicity in the sense of Koch's postulates has only been proven in a few cases .
In the vector, the spiroplasmas colonize numerous organs such as the digestive tract, the hemolymph , the salivary gland, the adipose tissue, the Malpighian vessels , the connective tissue, the nervous system and the ovaries . However, there are hardly any pathogenic influences. Reduced vitality, reduced survivability, hypertrophy of cells and cell nuclei and in some cases tissue lesions occur rarely .
In plants, however, the effect is often considerable. It occurred chlorosis on. The phloem changes degeneratively through severe disorganization. Affected cells change color and tend to hyperplasia . Necrotization and obliteration of phloem parts follow. In yellowed leaves, the palisade parenchyma recedes in favor of densely arranged sponge parenchyma, chloroplasts degenerate. Disturbances in the metabolism of the growth regulators lead to the lifting of growth inhibitions. For example, acropetal growth continues after flower formation or the induction of hibernation is prevented. Conversely, growth can also be inhibited, with consequences such as dwarfism, small leaves or small fruits.
When two types of Spiroplasma so-called was Male Killing observed. This means that the hosts no longer give birth to male offspring. This happens either through feminization of genetic males during sexual differentiation , through induction of parthenogenesis, or through death of the male offspring during embryogenesis . Male-killing has so far been associated almost exclusively with bacteria of the genus Wolbachia . Spiroplasma poulsonii , however, causes male killing in the fruit fly Drosophila willistoni and in two beetles, the two-point ladybird ( Adalia bipunctata ) and the Asian ladybird ( Harmonia axyridis ). Spiroplasma ixodetis has the same effect on ticks.
There is some controversial evidence of the role of spiroplasma in the etiology of transmissible spongiform encephalopathy (TSEs), particularly in the work of Frank Bastian. Other researchers have not been able to replicate these results , whereas the prion model for TSEs has now gained wide acceptance. A 2006 study also disproves the role of spiroplasma in hamster scrapie .
Internal system
The genus Spiroplasma has not yet been finally examined and the internal system is still subject to changes. A work from 2004 examines the 16S rDNA and tries to classify the genus. The following species seem to be reasonably certain:
- Spiroplasma citri ( serotype I-1): The acorn fruiting agent in citrus plants.
- Spiroplasma melliferum (serotype I-2): in honey bees
- Spiroplasma kunkelii , (serotype I-3): causative agent of maize upset
- Spiroplasma insolitum (serotype I-4): in rabbit ticks ( haemaphysalis ), on flowers and in butterflies
- Spiroplasma phoeniceum (serotype I): in the rose-colored Catharanthe ( Catharanthus roseus )
- Spiroplasma poulsonii (serotype II): in Drosophila flies - infected females have no male offspring
- Spiroplasma floricola (serotype III): on flowers and in cockchafer ( Melolontha )
- Spiroplasma apis (serotype IV): in honey bees
- Spiroplasma mirum (serotype V): in rabbit ticks ( Haemaphysalis )
- Spiroplasma ixodetis (serotype VI): in ticks - infected females have no male offspring
- Spiroplasma monobiae (serotype VII): in wasps
- Spiroplasma syrphidicola (serotype VIII-1): in hover flies of the genus Eristalis
- Spiroplasma chrysopicola (serotype VIII-2):
- Spiroplasma clarkii (serotype IX): Cotinus nitida in June beetle
- Spiroplasma culicicola (serotype X): in the mosquito Aedes sollicitans
- Spiroplasma velocicrescens (serotype XI): in the wasp Monobia quadridens
- Spiroplasma diabroticae (serotype XII): in the beetle Diabrotica undecimpunctata
- Spiroplasma sabaudiense (serotype XIII): in mosquitoes
- Spiroplasma corruscae (serotype XIV): in fireflies (Lampyridae) and horseflies (Tabanidae)
- Spiroplasma turonicum (serotype XVII): in horse horseflies
- Spiroplasma litorale (serotype XVIII): in horse horseflies
- Spiroplasma lampyridicola (serotype XIX): in fireflies
- Spiroplasma leptinotarsae (serotype XX): in the Colorado potato beetle ( Leptinotarsa decemlineata )
- Spiroplasma taiwanense (serotype XXII): in mosquitoes
- Spiroplasma gladiatoris (serotype XXIII): in horse horseflies
- Spiroplasma chinense (serotype XXIV): in the bindweed Calystegia hederacea
- Spiroplasma diminutum (serotype XXV): in mosquitoes
- Spiroplasma alleghenense (serotype XXVI): in the scorpion fly Panorpa helena
- Spiroplasma montanense (serotype XXXI): in horse horseflies
- Spiroplasma helicoides (serotype XXXII): in horse horseflies
literature
Unless otherwise stated, the information comes from this article:
- Franz Nienhaus: Viruses, Mycoplasma and Rickettsia . Ulmer, Stuttgart 1985, ISBN 3-8001-2536-6 .
Individual evidence
- ^ SL O'Neill, AA Hoffmann, JH Werren: Influential Passengers . Oxford University Press, Oxford 1997 (English).
- ↑ J. Hinrich Graf von der Schulenburg, Tamsin MO Majerus, Chorduraa M. Dorzhu, Ilia A. Zakharov, Gregory DD Hurst, Michael EN Majerus: Evolution of male-killing Spiroplasma (Procaryotae: Mollicutes) inferred from ribosomal spacer sequences . In: Journal of General and Applied Microbiology . tape 46 , 2000, pp. 95-98 , PMID 12483596 (English).
- ^ RH Leach, WB Mathews, R. Will: Creutzfeldt-Jakob disease. "Failure to detect spiroplasmas by cultivation and serological tests" . In: Journal of Neurological Science . tape 59 , no. 3 , 1983, p. 349-353 , PMID 6348215 (English).
- ↑ I. Alexeeva, EJ Elliott, S. Rollins, GE Gasparich, J. Lazar, RG Rohwer: Absence of Spiro plasma or Other Bacterial 16S rRNA Genes in Brain Tissue of hamster with scrapie . In: Journal of Clinical Microbiology . tape 44 , no. 1 , 2006, p. 91-97 , doi : 10.1128 / JCM.44.1.91-97.2006 , PMID 16390954 (English).
- Jump up ↑ Gail E. Gasparich, Robert F. Whitcomb, Deborah Dodge, Frank E. French, John Glass, David L. Williamson: The genus Spiroplasma and its non-helical descendants: phylogenetic classification, correlation with phenotype and roots of the Mycoplasma mycoides clade . In: International Journal of Systematic and Evolutionary Microbiology . tape 54 , no. 3 , 2004, doi : 10.1099 / ijs.0.02688-0 (English).
- ↑ C. Saillard, JC Vignault, JM Bové, A. Raie, JG Tully, DL Williamson, RF Whitcomb: Spiroplasma phoeniceum sp. nov., a new plant-pathogenic species from Syria . In: International Journal of Systematic and Evolutionary Microbiology . tape 37 , no. 2 , 1987, pp. 106-115 , doi : 10.1099 / 00207713-37-2-106 .
