Vanadocene dichloride
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
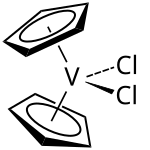
|
||||||||||||||||
| Crystal system |
monoclinic |
|||||||||||||||
| General | ||||||||||||||||
| Surname | Dichlorobis (η 5 -cyclopentadienyl) vanadium | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 10 H 10 Cl 2 V | |||||||||||||||
| Brief description |
green crystals |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 252.03 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
1.60 g cm −3 |
|||||||||||||||
| Melting point |
250 ° C (decomposition) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Vanadocene dichloride or, according to the IUPAC nomenclature, dichlorobis ( η 5 - cyclopentadienyl ) vanadium (IV) , is an organometallic compound from the family of metallocenes . The green solid is structurally identical to the titanocene dichloride . Vanadocene dichloride is a 17 electron complex and is paramagnetic due to its unpaired electron .
Extraction and presentation
Vanadocene dichloride was first produced by Geoffrey Wilkinson and Birmingham by reacting vanadium tetrachloride VCl 4 with cyclopentadienyl sodium NaC 5 H 5 in tetrahydrofuran .
For purification, the product was extracted with chloroform and hydrochloric acid and recrystallized with toluene .
properties
Vanadocene dichloride decomposes in the presence of moisture with the formation of hydrogen chloride .
use
Vanadocene dichloride is a common precursor for the production of bis (cyclopentadienyl) vanadium (IV) compounds. Vanadocene V (C 5 H 5 ) 2 is obtained by reducing vanadocene dichloride .
Biological importance
Like titanocene dichloride , vanadocene dichloride is also a potential candidate for cancer therapy and is therefore the subject of numerous studies. The mechanism is not yet clear, but there is evidence that it interacts with the protein transferrin .
Individual evidence
- ↑ a b c d e data sheet bis (cyclopentadienyl) vanadium (IV) dichloride from Sigma-Aldrich , accessed on June 16, 2014 ( PDF ).
- ↑ a b c data sheet bis (cyclopentadienyl) vanadium dichloride, 95% from AlfaAesar, accessed on June 18, 2014 ( PDF )(JavaScript required) .
- ↑ G. Wilkinson and JG Birmingham: Bis-cyclopentadienyl Compounds of Ti, Zr, V, Nb and Ta . In: Journal of the American Chemical Society . tape 76 , no. 17 , 1954, pp. 4281-4284 , doi : 10.1021 / ja01646a008 .
- ↑ Alaa S. Abd-El-Aziz: Inorganic and Organometallic Macromolecules. Springer Science & Business Media, 2007, ISBN 978-0-387-72947-3 , p. 227 ( limited preview in Google book search).
- ↑ Murthy MS, Rao LN, Kuo LY, Toney JH, Marks TJ: Antitumor and toxicologic properties of the organometallic anticancer agent vanadocene dichloride. In: Inorg. Chimica Acta . tape 152 , no. 2 , 1956, p. 117-124 , doi : 10.1016 / S0020-1693 (00) 83343-5 .

![{\ displaystyle \ mathrm {VCl_ {4} + \ 2 \ Na (C_ {5} H_ {5}) \ {\ xrightarrow [{}] {THF}} \ VCl_ {2} (C_ {5} H_ {5 }) _ {2} + \ 2 \ NaCl}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/4feecf8d938c032051dc398f3cd6a1a57a30d7b1)