Yttrium phosphate
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
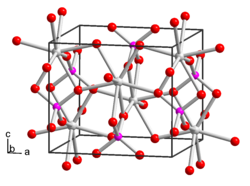
|
||||||||||||||||
| __ Y 3+ __ P 5+ __ O 2− | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Yttrium phosphate | |||||||||||||||
| other names |
Yttrium (III) phosphate |
|||||||||||||||
| Ratio formula | YPO 4 | |||||||||||||||
| Brief description |
colorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 183.88 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
|
|||||||||||||||
| Melting point |
1995 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Yttrium phosphate is an inorganic chemical compound of yttrium from the group of phosphates .
Occurrence
Yttrium phosphate occurs naturally in the form of the mineral xenotime and as a dihydrate as a Weinschenkit .
Extraction and presentation
Yttrium phosphate can be obtained by reacting yttrium (III) chloride with sodium phosphate at 200–1000 ° C or by reacting yttrium (III) nitrate with ammonium dihydrogen phosphate .
It is also possible to produce it by reacting yttrium oxide with diammonium hydrogen phosphate .
properties
Yttrium phosphate is a colorless solid that is partially soluble in phosphoric acid and slightly soluble in water. It has a crystal structure isotypic to that of zircon .
use
Yttrium phosphate is used as a catalyst and is a possible containment material for nuclear waste. Yttrium phosphate doped with cerium or bismuth shows luminescence in the UV range and can be used for tanning lamps.
Individual evidence
- ↑ a b c d e Jane E. Macintyre: Dictionary of Inorganic Compounds . CRC Press, 1992, ISBN 0-412-30120-2 , pp. 3704 ( limited preview in Google Book search).
- ↑ a b c data sheet yttrium (III) phosphate, ≥99.99% trace metals basis from Sigma-Aldrich , accessed on April 29, 2017 ( PDF ).
- ^ Bulletin of the National Research Council . National Academies, 1919, pp. 145 ( limited preview in Google Book search).
- ↑ a b minsocam.org: WEINSCHENKITE, YTTRIUM PHOSPHATE DIHYDRATE , accessed on May 16, 2014
- ^ DEC Corbridge: Phosphorus: Chemistry, Biochemistry and Technology, Sixth Edition . CRC Press, 2013, ISBN 1-4398-4088-1 , pp. 192 ( limited preview in Google Book search).
- ↑ Georg Brauer (Ed.), With the collaboration of Marianne Baudler a . a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6 , p. 1114.
- ↑ Ralf Alsfasser, Erwin Riedel: Modern Inorganic Chemistry . Walter de Gruyter, 2007, ISBN 3-11-019060-5 , p. 318 ( limited preview in Google Book search).


