Acitretin
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
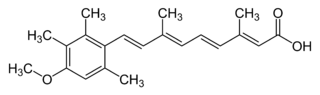
|
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Non-proprietary name | Acitretin | |||||||||||||||||||||
| other names |
(2 E , 4 E , 6 E , 8 E ) -9- (4-Methoxy-2,3,6-trimethylphenyl) -3,7-dimethylnona-2,4,6,8-tetraenoic acid ( IUPAC ) |
|||||||||||||||||||||
| Molecular formula | C 21 H 26 O 3 | |||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Drug information | ||||||||||||||||||||||
| ATC code | ||||||||||||||||||||||
| Drug class | ||||||||||||||||||||||
| Mechanism of action |
Control of cell growth and maturation of corneal cells |
|||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 326.4 g mol −1 | |||||||||||||||||||||
| Physical state |
firmly |
|||||||||||||||||||||
| Melting point |
228-230 ° C |
|||||||||||||||||||||
| pK s value |
4.2 |
|||||||||||||||||||||
| solubility |
0.00007 g l −1 in water at 25 ° C |
|||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Toxicological data | ||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Acitretin is a retinoid that is used as a drug in the treatment of psoriasis and other skin conditions .
chemistry
Acitretin is the non-proprietary name (INN) for (2 E , 4 E , 6 E , 8 E ) -9- (4-methoxy-2,3,6-trimethylphenyl) -3,7-dimethylnona-2,4,6, 8-tetraenoic acid (according to IUPAC ). It is an analogue of tretinoin (all- trans retinoic acid).
Mode of action
Acitretin is used in drugs against psoriasis, but is also used for cornification disorders or other skin diseases. Acitretin acts on and controls the growth of cells and the maturation of corneal cells. Acitretin also affects the immune responses that take place in the dermis . It is precisely these two mechanisms that are subject to changes in psoriasis. The active ingredient is used when an external treatment does not work satisfactorily.
Finished medicinal products
Acicutan (D), Neotigason (A, D, CH and others), Soriatane (USA, CDN)
Individual evidence
- ↑ a b c d e Data sheet Acitretin, ≥98.0% (HPLC) from Sigma-Aldrich , accessed on September 16, 2012 ( PDF ).
- ^ Entry on Acitretin at ChemicalBook , accessed September 14, 2012.
- ^ Entry on acitretin in the Hazardous Substances Data Bank , accessed September 14, 2012.
- ↑ a b Entry on acitretin in Flexikon , a Wiki of the DocCheck company , accessed on September 14, 2012.
- ^ Entry on acitretin in Pharmawiki , accessed on September 14, 2012.
- ↑ Acitretin at onmeda ( Memento from March 9, 2012 in the Internet Archive ). Retrieved September 14, 2012.