Ammonium thiosulfate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
2 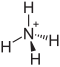 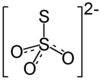
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Ammonium thiosulfate | |||||||||||||||
| other names |
ATS |
|||||||||||||||
| Molecular formula | (NH 4 ) 2 S 2 O 3 | |||||||||||||||
| Brief description |
white odorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 148.21 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
1.679 g cm −3 |
|||||||||||||||
| Melting point |
150 ° C (decomposition) |
|||||||||||||||
| solubility |
|
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Ammonium thiosulfate is a chemical compound from the group of ammonium salts and inorganic thiosulfates .
Extraction and presentation
Ammonium thiosulphate can be obtained by reacting ammonium sulphite with sulfur or sulphides in aqueous solutions (partly with ammonia), oxidation of hydrogen sulphide , through reaction of ammonium hydrogen carbonate with sulfur dioxide .
properties
Ammonium thiosulfate decomposes above 150 ° C, producing sulfur oxides, ammonia and nitrogen oxides.
use
Ammonium thiosulphate is used as a quick fixing salt (similar to sodium thiosulphate ) for the production of fixing baths in film technology, photo technology and X-ray technology and as a fertilizer.
Web links
Individual evidence
- ↑ a b c d e f g h i Entry on ammonium thiosulphate in the GESTIS substance database of the IFA , accessed on December 22, 2019 (JavaScript required)
- ↑ Data sheet ammonium thiosulfate (PDF) from Merck , accessed on March 17, 2010.
- ^ Karl Heinz Büchel, Hans-Heinrich Moretto, Peter Woditsch: Industrial Inorganic Chemistry. ISBN 978-3-527-29849-5 .
- ↑ Process for the production of ammonium thiosulfate (Patent.de)
- ↑ Safety data sheet (fld-friedland) ( page no longer available , search in web archives ) Info: The link was automatically marked as defective. Please check the link according to the instructions and then remove this notice. (PDF; 45 kB)


