Batrachotoxin
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
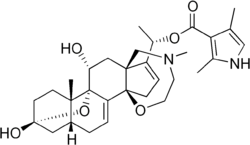
|
||||||||||
| General | ||||||||||
| Surname | Batrachotoxin | |||||||||
| other names |
Batrachotoxinin A-20- (2,4-dimethyl-1 H -pyrrole-3-carboxylate) |
|||||||||
| Molecular formula | C 31 H 42 N 2 O 6 | |||||||||
| Brief description |
non-crystalline solid |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 538.68 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| Toxicological data | ||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Batrachotoxins ( BTX ) (from ancient Greek βάτραχος bátrachos , German 'frog' and τοξικόν [φάρμακον] toxikón [phármakon] , German ' poison , with which the arrows are struck' ) are extremely potent neurotoxic steroid alkaloids ( pregnane derivatives) derived from the skin South American poison dart frogs of the genus Blattsteiger ( Phyllobates ). It has also been found in the skin and plumage of various New Guinea birds .

Scientific discovery of poisonous animals
The existence of poisonous animals had been known to the natives for many millennia, but batrachotoxin was not scientifically described in Colombian poison dart frogs until the 1960s. In the 1990s it was found that the skin and feathers of the two-color pitohui ( Pitohui dichrous ) living in New Guinea also contain batrachotoxin. Also Pitohui ferrugineus and Black Pitohui ( Pitohui nigrescens ) and Blaukappenflöter ( Ifrita kowaldi ) from New Guinea carry the poison in their feathers. The frogs, on the other hand, have lethal doses of the poison, particularly the terrible poison dart frog ( Phyllobates terribilis ), the amount of which could kill ten adult humans.
effect
Batrachotoxin has no effect on healthy skin, but causes severe, long-lasting pain, similar to a bee sting, with the smallest injury. Oral intake leads to poisoning only in pathological conditions of the gastrointestinal tract. They inhibit the inactivation of the sodium channels , so they are convulsive toxins . Touching the skin and feathers e.g. B. Using the lips causes tingling and numbness. The lethal dose for humans is estimated to be 1 to 2 µg / kg body weight. This makes the toxin about ten times stronger than tetrodotoxin . It is currently the most toxic steroid alkaloid known .
Source of poison
Neither frogs nor birds appear to be able to metabolize batrachotoxin . Since the frogs are poisonous in different regions and they lose their toxicity in captivity, it was assumed that the animals ingest and accumulate the poison with their food . By questioning Papuan natives, beetles of the genus Choresine from the family Melyridae were found to be a source of poison ; related beetles are also found in Colombia.
Human handling of batrachotoxins
In western Colombia use Noanamá-Chocó - and Emberá Chocó -Indianer batrachotoxin as a poison for blowguns .
The poisonous birds of New Guinea are traditionally carefully skinned by the indigenous population before consumption.
An antidote to batrachotoxins is the paralytic tetrodotoxin , the poison of the puffer fish that blocks the sodium channels on the nerve axon.
Individual evidence
- ↑ a b c Entry on batrachotoxins. In: Römpp Online . Georg Thieme Verlag, accessed on June 20, 2014.
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Entry on batrachotoxin in the ChemIDplus database of the United States National Library of Medicine (NLM) .
- ^ Wilhelm Pape , Max Sengebusch (arrangement): Concise dictionary of the Greek language. 3rd edition, 6th impression, Vieweg & Sohn, Braunschweig 1914. 1914, accessed on July 27, 2020 .
- ^ Wilhelm Pape , Max Sengebusch (arrangement): Concise dictionary of the Greek language. 3rd edition, 6th impression, Vieweg & Sohn, Braunschweig 1914. 1914, accessed on July 27, 2020 .
- ^ A b c d John Tidwell (2001): The intoxicating birds of New Guinea. ( Memento from July 9, 2015 in the Internet Archive ) (PDF) In: ZooGoer. Vol. 30, No. 2., 2001.
- ↑ Stephanie Greenman Stone, Pat Kilduff: New Research Shows that Toxic Birds and Poison-dart Frogs Likely Acquire their Toxins from Beetles. ( Memento of December 3, 2012 in the Internet Archive ) Newsroom of the California Academy of Sciences, article dated October 12, 2004.
- ↑ John P. Dumbacher, Avit Wako, Scott R. Derrickson, Allan Samuelson, Thomas F. Spande, John W. Daly: Melyrid beetles (Choresine): A putative source for the batrachotoxin alkaloids found in poison-dart frogs and toxic passerine birds . (PDF) In: PNAS 101, No. 45, 2004, pp. 15857–15860, doi : 10.1073 / pnas.0407197101 .
- ↑ Bethany Halford: Birds and beetles: A toxic trail. In: Chemical & Engineering News 82, No. 45, 2004, p. 17.
Web links
- Dumbacher, JP et al. (2004): Melyrid beetles (Choresine): a putative source for the batrachotoxin alkaloids found in poison-dart frogs and toxic passerine birds. In: Proc. Natl. Acad. Sci. USA Vol. 101, pp. 15857-15860. PMID 15520388 PDF
- Protection from its own poison: the golden poison dart frog's survival trick , deutschlandfunk.de , 5 September 2017.
