Dodecahedran
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||
| General | ||||||||||
| Surname | Dodecahedran | |||||||||
| Molecular formula | C 20 H 20 | |||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 260.36 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| density |
1.434 g cm −3 |
|||||||||
| Melting point |
430 ° C |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Dodecahedrane is a molecule from the group of isocyclic hydrocarbons and has the empirical formula C 20 H 20 .
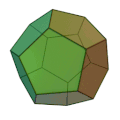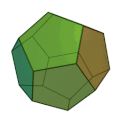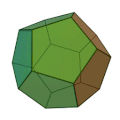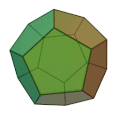
The molecule has the shape of a dodecahedron - a platonic solid - and thus belongs to the group of platonic hydrocarbons . Each carbon atom is connected to three other carbon atoms and one hydrogen atom . Thus, five carbon atoms always form one side of the 12-sided polyhedron . The dodecahedrane was first synthesized in 1982 by Leo A. Paquette . The compound crystallizes in a cubic crystal lattice.
Dodecahedran, dynamic presentation. All edges are C – C bonds, methine groups (CH groups) form the corners
Dodecahedran dome model
synthesis
The route to dodecahedrane taken by Leo A. Paquette made the initial synthesis possible, but subsequently proved to be too expensive to provide multi-gram quantities of dodecahedrane and to investigate the chemistry of the dodecahedrane in detail. This was only made possible by the pagodan route to the dodecahedran worked out in the Horst Prinzbach working group . The route is named after the hydrocarbon pagodan (C 20 H 20 ), since in its original variant dodecahedrane was obtained by catalyzed gas-phase isomerization of pagodan. A summary of the route that has been optimized over several years in its final version - which now still runs via pagodan derivatives but avoids the direct isomerization of pagodan to dodecahedrane, which can only be achieved in low yields - can be found in the following scheme.
The synthesis begins with isodrine (1), which was previously used as an insecticide . In the course of the synthesis, some less known reaction steps are carried out.
The following are to be emphasized:
- Dyotropic H-transfer (3) → (4)
- [2 + 2] / [6 + 6] photocycloaddition (9) → (10)
- transannular hydride transfer with entry of a nucleophile (17) → (18)
- Use of a "naked" fluoride as a sterically lean, strong base (18) → (19)
Individual evidence
- ↑ a b c Bertau, M .; Wahl, F .; Weiler, A .; Scheumann, K .; Woerth, J .; Keller, M .; Prinzbach, H .: From Pagodanes to Dodecahedranes - Search for a Serviceable Access to the Parent (C 20 H 20 ) Hydrocarbon , in: Tetrahedron , 53 (1997), pp. 10029-10040, doi : 10.1016 / S0040-4020 (97 ) 00345-1 .
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Entry on platonic hydrocarbons. In: Römpp Online . Georg Thieme Verlag, accessed on June 19, 2014.
- ↑ Ternansky, RJ; Balogh, DW; Paquette, LA: Dodecahedrane , in: J. Am. Chem. Soc. , 104 (1982), pp. 4503-4504, doi : 10.1021 / ja00380a040 .
- ↑ Wolf Dieter Fessner, Gottfried Sedelmeier, Paul R. Spurr, Grety Rihs, and H. Prinzbach. "Pagodane": the efficient synthesis of a novel, versatile molecular framework. In: Journal of the American Chemical Society , 1987, 109 (15), pp. 4626-4642, doi : 10.1021 / ja00249a029 .
- ↑ Wolf-Dieter Fessner, Bulusu ARC Murty, Horst Prinzbach (1987), The Pagodane Route to Dodecahedranes - Thermal, Reductive, and Oxidative Transformations of Pagodanes Angewandte Chemie International Edition in English , Volume 26, Issue 5, pp. 451–452, doi : 10.1002 / anie.198704511 .
- ↑ Wolf-Dieter Fessner, Bulusu ARC Murty, Jürgen Wörth, Dieter Hunkler, Hans Fritz, Horst Prinzbach, Wolfgang D. Roth, Paul von Ragué Schleyer, Alan B. McEwen, Wilhelm F. Maier (1987), Dodecahedranes from [1.1. 1.1] Pagodanes . Angewandte Chemie International Edition in English , Volume 26, Issue 5, pp. 452–454, doi : 10.1002 / anie.198704521 .
- ^ H. Prinzbach, F. Wahl, A. Weiler, P. Landenberger, J. Woerth, LT Scott, M. Gelmont, D. Olevano, F. Sommer, B. von Issendorff: C20 Carbon Clusters: Fullerene - Boat - Sheet Generation, mass selection, PE characterization. Chem. Eur. J. , 2006, 12, pp. 6268-6280, doi : 10.1002 / chem.200501611 .