Folinic acid
| This item has been on the quality assurance side of the editorial chemistry entered. This is done in order to bring the quality of the articles on the subject of chemistry in terms of form and content to the level desired in Wikipedia. We are grateful for your help , please take part in the discussion ( new entry ) or revise the article accordingly. |
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
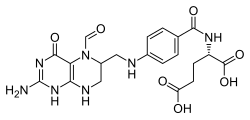
|
|||||||||||||||||||
| Simplified structural formula without complete stereochemistry | |||||||||||||||||||
| General | |||||||||||||||||||
| Non-proprietary name | Folinic acid (modified non-proprietary name) | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 20 H 23 N 7 O 7 | ||||||||||||||||||
| Brief description |
pale yellow crystals (folinic acid trihydrate); pale beige powder (calcium salt pentahydrate) |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| Drug information | |||||||||||||||||||
| ATC code | |||||||||||||||||||
| Drug class |
Detoxifying agent for treatment with cytotoxic drugs |
||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 473.44 g · mol -1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
248-250 ° C (dec.); 240–250 ° C (decomposition) (calcium salt pentahydrate) |
||||||||||||||||||
| pK s value |
3.3 |
||||||||||||||||||
| solubility |
Hardly soluble in water |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| Toxicological data |
1063 mg kg −1 ( LD 50 , rat , ip , Folinic acid calcium salt pentahydrate) |
||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Folinic acid ( FA ), also citrovorum factor ( CF ) or leucovorin ( LV ), short- CHO-FH 4 is the 5- formyl - derivative of tetrahydrofolic acid , which as the active form of folic acid in various metabolic processes ( nucleotide synthesis , amino acid metabolism involved) is. Folinic acid is used medicinally
- in combination with 5-fluorouracil in the treatment of colon cancer ,
- to reduce or counteract the cytotoxic effects of drugs from the group of so-called folic acid antagonists ,
- in low doses for the treatment of folic acid deficiencies that cannot be remedied by dietary measures.
chemistry
Folinic acid has two stereocenters, so there are four isomers in total. Most finished medicinal products contain a mixture of folinic acid diastereomers . The calcium salt (calcium folinate), its hydrate (calcium folinate pentahydrate) and the disodium salt (sodium folinate) are used.
Meanwhile, however, preparations with the pure, biologically active form, the L - (-) - isomer called levofolic acid, are available. The calcium salt is also used here.
Pharmacological effect and areas of application
Combination therapy with 5-fluorouracil
Folinic acid is used in synergistic combination with the cytostatic 5-fluorouracil (5-FU) in chemotherapy for the treatment of colon cancer and other tumors. Folinic acid binds to the enzyme thymidylate synthase and thereby lowers the intracellular thymidylate concentration, which increases the cytostatic effect of 5-FU.
“Leukovorin Rescue” during therapy with high-dose methotrexate
Folinic acid is used as an antidote in methotrexate (MTX) therapies. Methotrexate acts as a folic acid antagonist and competitively and reversibly inhibits the enzyme dihydrofolate reductase (DHFR). In the case of high-dose methotrexate administration, a so-called "leukovorin rescue" must take place, i. H. The effect of the methotrexate must be antagonized by regular administration of Leukovorin, as otherwise severe bone marrow and mucous membrane toxicity ( mucositis ) occurs. In practice, the procedure is usually that methotrexate is administered as an infusion over approx. 4 hours and the Leukovorin Rescue begins 24 hours after the infusion (an infusion with Leukovorin every 6 hours) until the methotrexate level is reached has fallen below a critical limit (<0.1 µM ).
Antidote to trimetrexate, trimethoprim and pyrimethamine
Similar to therapy with high-dose methotrexate, folinic acid is also indicated as an antidote in high-dose therapy with the folic acid antagonists trimetrexate , trimethoprim and pyrimethamine .
Treatment of folic acid deficiency
If a vitamin B 12 deficiency has been excluded from the differential diagnosis, folinic acid can be used in low doses (maximum 15 mg per day) to treat folic acid deficiency. It is also used to treat cerebral folate deficiency .
Individual evidence
- ↑ a b c Entry on folinic acid. In: Römpp Online . Georg Thieme Verlag, accessed on July 2, 2019.
- ↑ a b c d Data sheet Folinic acid calcium salt hydrate from Sigma-Aldrich , accessed on April 2, 2011 ( PDF ).
- ^ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals , 14th Edition (Merck & Co., Inc.), Whitehouse Station, NJ, USA, 2006; P. 723, ISBN 978-0-911910-00-1 .
- ^ The Merck Index. An Encyclopaedia of Chemicals, Drugs and Biologicals . 9th edition, 1976, ISBN 0-911910-26-3 , p. 544.
- ↑ External identifiers or database links to calcium folinate: CAS number: 1492-18-8, EC number: 216-082-8, ECHA InfoCard: 100.014.621 , PubChem : 15150 , ChemSpider : 14421 , Wikidata : Q27077063 .
- ↑ External identifiers or database links for calcium folinate pentahydrate : CAS number: 6035-45-6, EC number: 622-950-0, ECHA InfoCard: 100.151.674 , PubChem : 20835958 , ChemSpider : 20137742 , Wikidata : Q27287765 .
- ↑ External identifiers or database links for sodium folinate: CAS number: 163254-40-8, PubChem : 71587560 , ChemSpider : 32702403 , Wikidata : Q27260173 .
- ↑ External identifiers or database links for levofolic acid : CAS number: 68538-85-2, PubChem : 149436 , ChemSpider : 131714 , DrugBank : DB11596 , Wikidata : Q192464 .
- ↑ External identifiers or database links for calcium levofolinate: CAS number: 80433-71-2, EC number: 801-530-2, ECHA InfoCard: 100.228.231 , PubChem : 9892583 , ChemSpider : 8068253 , Wikidata : Q27266563 .
