Hoelit
| Hoelit | |
|---|---|
| Yellow needle-like hoelite crystals with sulfur (image width: 10 mm) | |
| General and classification | |
| chemical formula | (C 6 H 4 ) 2 (CO) 2 |
|
Mineral class (and possibly department) |
Organic mineral |
|
System no. to Strunz and to Dana |
10.CA.15 ( 8th edition : IX / B.02) 04/50/02/01 |
| Crystallographic Data | |
| Crystal system | monoclinic, pseudo-orthorhombic |
| Crystal class ; symbol | monoclinic prismatic 2 / m |
| Space group | P 2 1 / a |
| Lattice parameters |
a = 15.810 Å ; b = 3.942 Å; c = 7.895 Å β = 102.72 ° |
| Formula units | Z = 2 |
| Physical Properties | |
| Mohs hardness | not defined |
| Density (g / cm 3 ) | 1.42 |
| Cleavage | Well |
| colour | yellow to yellow-green |
| Line color | pale yellow |
| transparency | translucent |
| shine | Please complete |
| Crystal optics | |
| Refractive indices |
n α = 1.75 n β = 1.75 n γ = 2.0 |
| Optical character | biaxial positive |
Hoelite (chemically anthraquinone ) is a rarely occurring mineral from the mineral class of organic compounds . It crystallizes in the monoclinic crystal system with the chemical composition (C 6 H 4 ) 2 (CO) 2 and develops fine-needle, sometimes pseudo-orthorhombic crystals in the millimeter range from yellow to yellow-green in color.
Etymology and history
The mineral was discovered by Adolf Hoel , after whom it was named, in 1922 in pyramids on Svalbard .
classification
In the old system of minerals according to Strunz (8th edition) , the Hoelit belongs to the division of nitrogen-free hydrocarbons . Since the new edition of Strunz's mineral system (9th edition) according to Strunz , the class of organic minerals has been divided differently and the mineral can now be found in the subdivision “10.CA.” of the department “Various organic minerals”.
The systematics of minerals according to Dana sorts the Hoelit into the department of "salts of organic acids with different formulas".
Education and Locations
Hoelite forms in burning coal deposits , often together with ammonia and / or sulfur .
So far, the mineral has only been found at Libušín and Radvanice v Čechách (Okres Trutnov) in Bohemia (Czech Republic), apart from its type locality Spitzbergen ; as well as in Saxony (Freital) in Germany.
Crystal structure
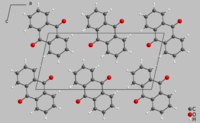 Crystal structure of hoelite |
|
| Crystal system | monoclinic |
| Space group | |
|
Lattice parameter (unit cell ) |
a = 15.810 Å b = 3.942 Å c = 7.895 Å β = 102.72 ° |
| Number (Z) of the formula units |
Z = 2 |
Hoelite crystallizes in the monoclinic crystal system in the space group P 2 1 / a with the lattice parameters a = 15.810 Å , b = 3.942 Å, c = 7.895 Å and β = 102.72 ° as well as two formula units per unit cell . The crystal structure consists of individual anthraquinone - molecules , which Van der Waals interactions are held together.
Individual evidence
- ↑ a b c d e American Mineralogist Crystal Structure Database - Hoelite (Engl., 1967)
- ↑ a b c d e Webmineral - Hoelite (engl.)
- ↑ a b c Mineral data sheet - Hoelite (English, PDF 58.6 kB).
- ↑ Webmineral - New Dana Classification of Organic Minerals .
- ↑ MinDat - Hoelite (Eng.).
Web links
- Mineral Atlas: Hoelit (Wiki)


